
안녕하세요 보스턴 임박사입니다.
Kyverna Therapeutics는 Wastlake Village BioPartners에 의해 설립된 회사입니다.
BIOTECH (82) – Westlake Village BioPartners VC
Kyverna Therapeutics는 Gilead와 전략적 제휴를 해서 $570 Million 규모의 Deal을 초기부터 가지고 출범을 했습니다.
Series B는 $145 Million을 했습니다.
Kyverna Closes Oversubscribed $145M Series B Financing – Precision Medicine Online 8/3/2023
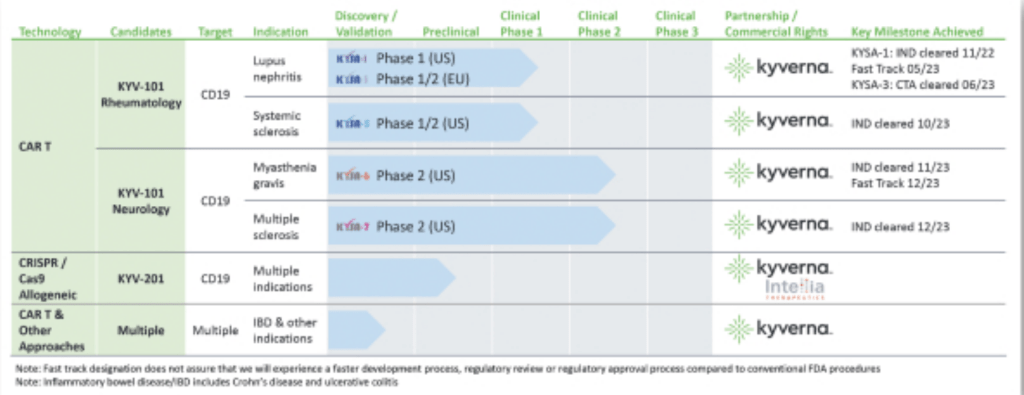
In testing KYV-101 in a Phase I/II study involving 20 cancer patients, the company noted the cell therapy caused fewer cytokine-driven side effects, such as immune effector cells-associated neurotoxicity syndrome, and reduced immunogenicity resulting in better cell persistence at one month. Based on this, Kyverna believes KYV-101 may work particularly well as a treatment for autoimmune diseases and obtained worldwide licenses from the National Institutes of Health to use the same cell therapy construct to make autologous and allogeneic versions of treatments in this setting.
In July, the firm began studying KYV-101, the autologous version, in lupus nephritis in the US in a Phase I trial. There is also a Phase I/II trial of KYV-101 ongoing in Germany. KYV-201 is the allogeneic version of the anti-CD19 CAR T-cell construct that Kyverna is studying as a treatment for lupus nephritis and other B cell-driven autoimmune diseases.
Kyverna Therapeutics의 기술은 NIH의 2020년 Nature Medicine 논문에 근거합니다.
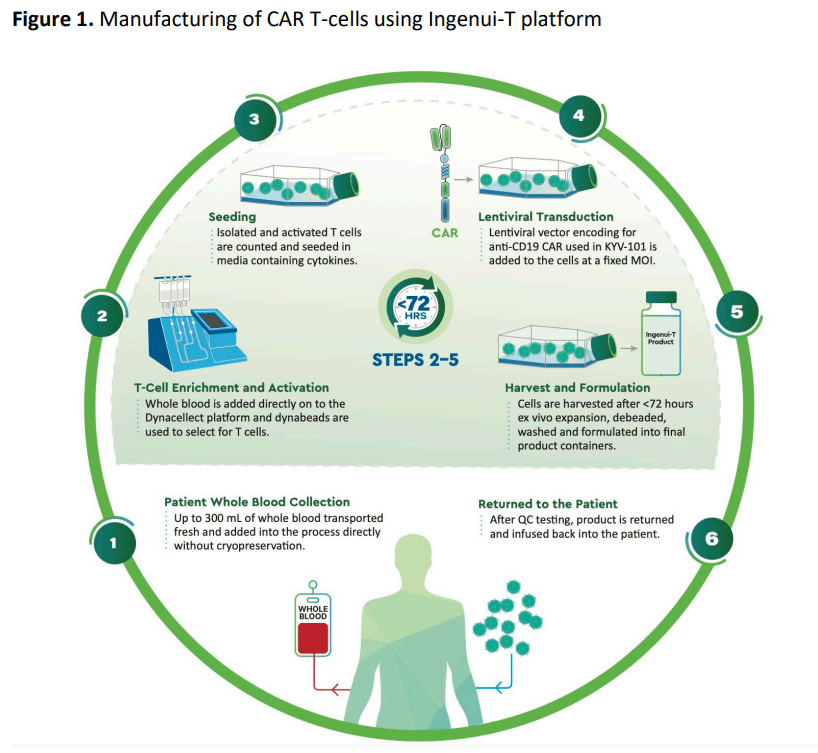
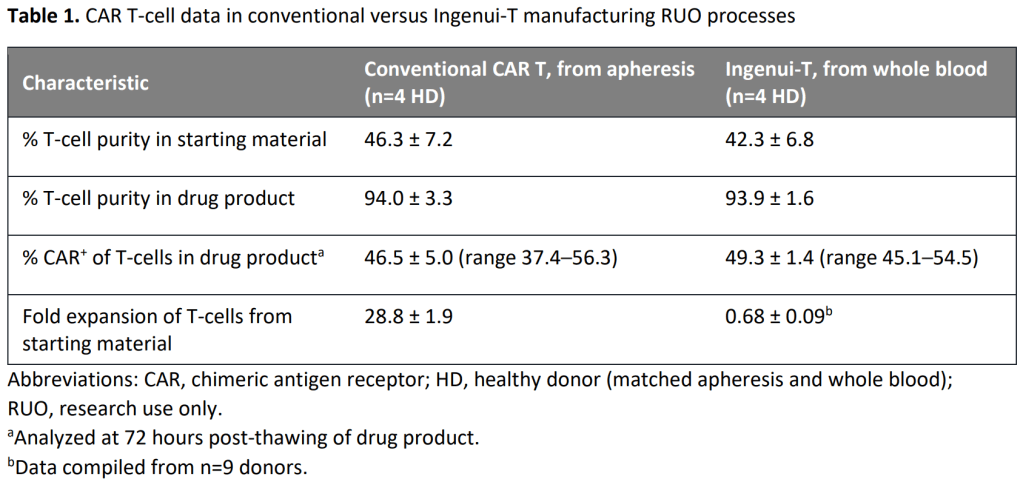
Ingenui-T platform은 기존 CAR-T 치료제 생산에서 필수적인 Apheresis step을 하지 않고 Whole blood를 <300 mL 채취한 후 72시간 (3일) 이내에 T-cell enrichment/activation 부터 Harvest/Formulation 까지 마치는 것을 핵심적인 Manufacturing Advantage로 하는 기술입니다.
Kyverna: 3-day process a ‘gamechanger’ for CAR-T – BioProcess International 1/29/2024
본래 IPO 규모는 $182 Million이었는데 실제로는 $319 Million IPO를 하게 된 것입니다.
아래는 Kyverna Therapeutics의 IPO를 위해 SEC에 등록한 S-1 Form입니다.
CAR-T를 Autoimmune disease에 이용하는 임상시험은 Kyverna Therapeutics가 처음인 것으로 알고 있는데요 어떤 결과를 얻을지 궁금합니다.
