안녕하세요 보스턴 임박사입니다.
Gilead Sciences는 HIV, HBV, HCV와 같은 Virology 분야의 세계최강자이고 Oncology 분야에서도 Yeskarta, Tecatus, Trodelvy, Zydelic 같은 항암제를 비롯한 다양한 Oncology Pipeline을 가지고 있습니다.
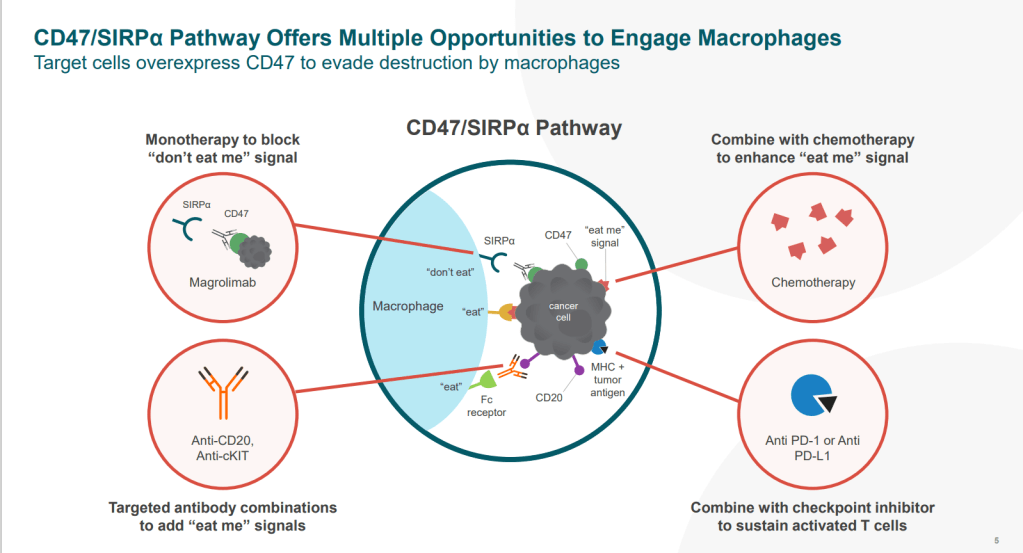
이 중에서 CD47-SIRPα Blocker인 Magrolimab (Hu5F9-G4)에 대해 얘기해 보려고 합니다.
Forty Seven Inc. 는 2015년에 Stanford University의 Irving Weissman교수의 CD47의 Immune evasion role에 대한 연구를 바탕으로 설립된 Biotech입니다.

How Irv Weissman learned to figure things out – Stanford Medicine News Center 1/22/2016
Company founder Irv Weissman said, “Targeting CD47 integrates the adaptive and innate immune systems, creating synergy with existing cancer-specific antibodies like rituximab, cetuximab and trastuzumab through ADCP, and potentially with T-cell checkpoint inhibitors through cross-presentation. We are grateful to the California Institute for Regenerative Medicine (CIRM) for funding the preclinical studies and the current solid tumor clinical trial at Stanford, and to Ludwig Cancer Research for funding much of the research.”
“The founders, Irv Weissman, Ravi Majeti, Mark Chao and Jens Volkmer, have studied the CD47 pathway extensively since they initially identified it as a cancer target in two papers published in 2009,” commented Chris Schaepe, Partner at Lightspeed Venture Partners. “The company’s scientific founders have done an outstanding job of advancing Hu5F9-G4 into two Phase 1 clinical trials in patients with relapsed or refractory solid tumors or acute myeloid leukemia (AML).”
Forty Seven raises another $75M to expand I-O trial slate – Fierce Biotech 10/18/2017
Forty Seven has the means to expand beyond its current slate of five phase 1b and phase 1b/2 clinical trials. The new trials will pair lead candidate Hu5F9-G4 to T-cell checkpoint inhibitors.
The combination could unleash a powerful two-front immune system attack on tumors. Hu5F9-G4 is designed to do for macrophages—white blood cells that gobble up other cells—what Merck’s Keytruda and Bristol-Myers Squibb’s Opdivo do for T cells. On paper, the combination will take the breaks off the anti-tumor activities of both T cells and macrophages.
Forty Seven, Inc. $112.6 Million Initial Public Offering – Davis Polk 7/2/2018
Stanford doctor will get $191 million in cancer-fighting biotech deal – Los Angeles Times 3/6/2020
Gilead Sciences는 2020년 3월에 Forty Seven을 2배 premium인 $4.9 Billion으로 인수합니다.
Gilead announced Monday it will acquire Forty Seven for $95.50 a share, almost double its closing price the preceding Thursday, before Bloomberg News reported on the potential for a deal. Co-founder Majeti’s stake is worth about $120 million. Forty Seven is the fourth company started by Weissman, who also served on the founding scientific advisory board of Amgen Inc.
In December, Forty Seven went some way toward validating its approach by revealing 50% or more of the myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML) patients who took magrolimab and Celgene’s Vidaza had a complete response. The data triggered a surge in Forty Seven’s stock, which went from below $15 to above $40 in two weeks. Now, the stock has climbed higher again—far higher. Gilead’s accepted bid values Forty Seven at $95 a share. For most of its time on public markets, Forty Seven has traded below $20, dipping as low as $6 back in October. Until Bloomberg broke news of Gilead’s interest in Forty Seven last week, the stock had never breached $50. The hefty premium will bag Gilead a drug that is being tested in a handful of indications. In addition to the aforementioned phase 2 trials in MDS and AML, Forty Seven is testing magrolimab in patients with non-Hodgkin lymphoma, diffuse large B-cell lymphoma and three solid tumor types, namely colorectal, ovarian and bladder.
Gilead Science-Forty Seven Deal은 Daniel O’Day가 CEO가 된 후 (2019년 3월) 가장 처음 한 M&A 였습니다.
인수한지 반년만에 Magrolimab은 FDA Breakthrough Therapy Designation을 받았습니다.
“The Breakthrough Therapy designation recognizes the potential for magrolimab to help address a significant unmet medical need for people with MDS and underscores the transformative potential of Gilead’s immuno-oncology therapies in development,” said Merdad Parsey, MD, PhD, Chief Medical Officer, Gilead Sciences. Magrolimab is currently being studied in the double-blind, placebo-controlled, randomized Phase 3 ENHANCE trial in previously untreated higher risk MDS. The trial will evaluate the safety and efficacy of magrolimab, in combination with azacitidine, as measured by CR and duration of CR.
하지만 2년이 채 지나지 않아서 환자가 사망하면서 MDS와 AML은 Partial Clinical Hold를 받게 되었습니다.
그리고 1년이 조금 지난 후 결국 MDS는 임상이 중단되었습니다.
그리고 1달이 지나서 FDA가 AML 임상3상에 대해서 다시 Partial Clinical Hold를 걸었습니다.
Abbvie/I-Mab의 anti-CD47 antibody와 Celgene의 anti-CD47 antibody는 임상 1상에서 중단된 바가 있습니다. Gilead의 Magrolimab은 anti-CD47 antibody 중 가장 최종단계 임상까지 간 케이스였습니다.
Gilead Axes Phase III Magrolimab Trial Based on Disappointing Data – Biospace 9/27/2023
While the anti-CD47 antibody was seen as a potential treatment for blood cancers, so far it has not proven very effective in the clinic. After inking a $2 billion deal in 2020, Chinese biotech I-Mab and partner AbbVie decided to call it quits on a Phase Ib trial of the former’s lemzoparlimab. The antibody was being tested in MDS and AML in combination with azacitidine and venetoclax.
This week, AbbVie made the decision to cut its losses and terminate its licensing and collaboration agreement with I-Mab for lemzoparlimab. I-Mab’s SEC filing called it a “strategic decision.” AbbVie is out the $200 million upfront it already paid out to I-Mab. Celgene, once considered a front runner for the anti-CD47 monoclonal antibody market, terminated its Phase I trial of CC-90002 in 2018 after preliminary data for AML and MDS did “not offer a sufficiently encouraging profile for further dose escalation/expansion.”
그리고 반년만에 Gilead Sciences는 모든 혈액암의 임상을 중단했습니다.
Independent data monitoring committee (IDMC)에서 AML Phase 3 ENHANCE-3 trials를 중단하도록 요청했고요 이로써 모든 혈액암 임상3상은 중단시켰지만 당시 고형암 임상은 2상으로 내려서 계속 진행할 생각이라고 밝혔습니다.
Gilead announced that the phase 3 ENHANCE-3 study is being discontinued in acute myeloid leukemia based on the recommendation of the independent data monitoring committee that had been reviewing top-line data from a planned interim analysis, according to a Wednesday release. The analysis showed an increased risk of death when adding magrolimab to the chemotherapy azacitidine and Genentech/AbbVie’s Venclexta, as well as futility. The deaths were caused by infections and respiratory failure, Gilead said. The FDA has now placed a full clinical hold on the anti-CD47 antibody in myelodysplastic syndromes (MDS) and AML, plus related expanded access programs.
Gilead will end all development of the therapy in blood cancer, setting the asset firmly back to phase 2 with tests in breast, lung and gastrointestinal cancers, and other solid tumors. The company is reviewing safety data across all the ongoing solid tumor trials and plans to provide an update soon.
그리고 1주일이 지난 이번주에 고형암 임상2상도 Partial Clinical Hold 상태가 된 것을 발표했습니다.
고형암의 임상시험도 최종적으로 중단이 될지 아니면 지속할 수 있을지 좀더 지켜봐야 할 것 같습니다.
