
Picture from https://www.lifescienceleader.com/doc/krystal-biotech-s-side-business-gene-based-aesthetics-0001
안녕하세요 보스턴 임박사입니다.
Krystal Biotech은 저에게 Biotech에 대한 Out-of-box concept을 일깨워주는 회사입니다. 사진에 보여드리는 것과 같이 이 회사는 부부가 창업하고 펀딩해서 Nasdaq IPO를 하고 결국 FDA 승인까지 단기간에 일구기까지 신기하기도 하고 배워볼 마음에 이 글을 씁니다.
How Krystal Biotech Went From Founding To IPO In 18 Months – Smart Business Dealmakers 8/28/2019
The Pittsburgh-based company, which Krishnan co-founded with his wife and COO Suma Krishnan, is working to develop to develop treatments for rare, orphan skin diseases caused by the absence of, or a mutation in, a single gene.
“We were humming along,” he says. “We weren’t really seriously thinking of going public in 2017. We thought maybe it would be a 2018, 2019 type thing. But we had some good interest from investors on Wall Street, ones that had been in biotech and tend to be patient and long term and let the story play out.”
Krishnan was bitten by the biotech bug early in his career, when his company got a drug approved and he heard stories from friends about their children being able to take the drug and feel better.
“The real goal is not as much to go IPO, it’s to get a good treatment for a disease approved,” he says.
The Krishnans self-funded the business in April 2016. They took on two rounds of corporate venture capital, raising more than $18 million, before going public in September 2017. As a public company, Krystal has done two additional rounds of financing, a private placement in 2018 and an additional public offering in June of this year, raising another $200 million in total.
The VC market provides value in terms of guidance and experience, but in exchange they take a lot of equity, Krishnan says. That means many times, entrepreneurs, after a couple of rounds of financing, realize they’re not the bosses anymore.
“The ownership shifts from the entrepreneur to the venture capitalists pretty quickly, and then the direction and a lot of decisions do not solely exist in the hands of the entrepreneur,” he says.
“In our case, given my prior record, it was a conscious decision to stay away and try and keep as much of the ownership in the company as possible,” he says.
Obviously, the downside is it’s a lot of work to keep up with the required procedures and processes. That’s why he recommends figuring out your business strategy first as a private company. Then, when you go public, it’s more about execution of a clear marching order.
Krystal was fortunate that its two funding rounds as a public company were able to be done on the back of good data.
Krystal Biotech nets $42.4mm in oversubscribed IPO – Citeline 9/22/2017
전임상 결과는 아래 논문에 보고를 했습니다.
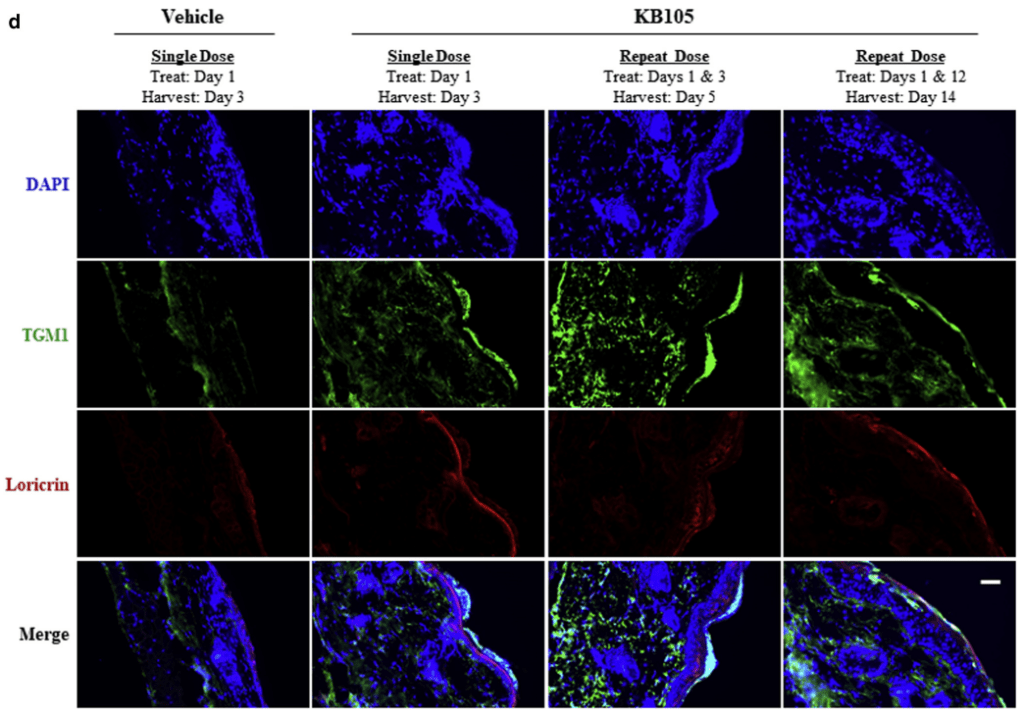
Krystal Biotech의 Vijuvek (beremagene geperpavec)의 임상 1상과 2상 결과는 Nature Medicine에 보고하고 임상3상 결과는 New England Journal of Medicine에 발표했습니다.
In the Phase 1 and 2 study, matched wounds were evaluated in nine RDEB patients receiving topical B-VEC or placebo repeatedly over 12 weeks. Primary and secondary mechanistic and clinical endpoints were met. No Grade 2 or above B-VEC-related adverse events, vector shedding, or systemic drug exposure were noted.
In this GEM-3 trial of 31 patients, complete wound healing at 6 months occurred in 67.4% of B-VEC wounds compared to 21.6% for placebo (difference, 45.8 percentage points; 95% confidence interval [CI], 23.6 to 68.0; p=0.002). Complete wound healing at 3 months occurred in 70.6% of the wounds exposed to B-VEC as compared with 19.7% of those exposed to placebo (difference, 51.0 percentage points; 95% CI, 29.3 to 72.6; p=0.0005).
The Company received US Food and Drug Administration (FDA) filing acceptance of its Biologics License Application (BLA) for B-VEC. The BLA was granted Priority Review designation and the Prescription Drug User Fee Act action date is February 17, 2023. The Company has filed the Marketing Authorization (MA) application with the European Medical Agency (EMA) and is currently working closely with the EMA through the MA validation process.
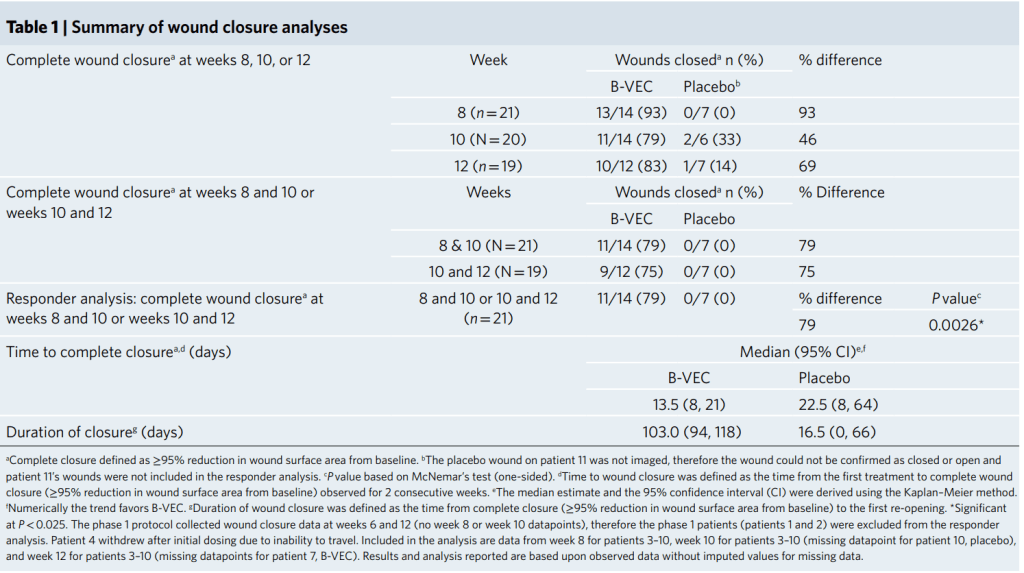
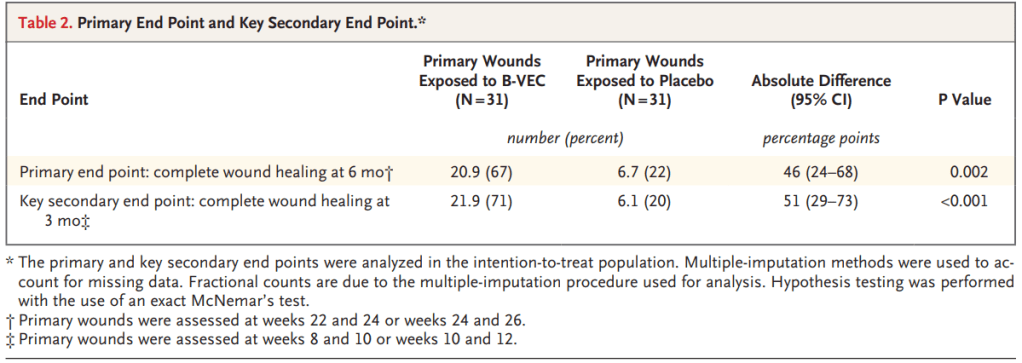
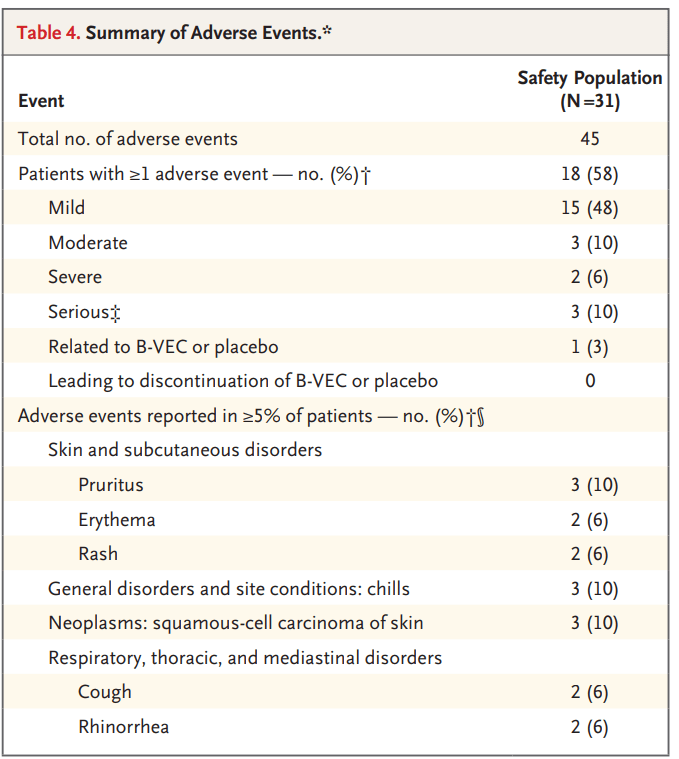
he FDA approval of Vyjuvek is based on two clinical studies. The GEM-1/2 trial was an intra-patient, open label, single center, randomized, placebo-controlled study showing that repeat topical applications of Vyjuvek were associated with durable wound closure, full-length cutaneous COL7 expression, and anchoring fibril assembly with minimal reported adverse events. The GEM-3 trial was an intra-patient, double-blinded, multi-center, randomized, placebo-controlled study that met both its primary endpoint of complete wound healing at six months and its key secondary endpoint of complete wound healing at three months. Vyjuvek was well tolerated with no drug-related serious adverse events or discontinuations due to treatment-related events.
“Data from our GEM-1/2 trial and our GEM-3 trial, published in Nature Medicine and the New England Journal of Medicine, respectively, demonstrated the strength of both studies showing that Vyjuvek safely and effectively improved wound healing,” said Suma Krishnan, president, Research & Development, Krystal Biotech.
이외에도 Krystal Biotech은 Jeune Aesthetics라는 subsidiary를 만들어서 KB301이라는 intradermal aesthetic injection을 개발하는 회사를 만들었습니다.
Krystal Biotech’s Side Business: Gene-Based Aesthetics – Life Science Leader 11/1/2023
In 2019, Krystal launched a subsidiary called Jeune Aesthetics to develop new applications of its STAR-D platform for the aesthetics market. Its first development candidate, called KB301, uses Krystal’s HSV vector and mechanism of action to deliver a COL31a transgene via intradermal injection, with the goal of boosting the skin’s ability to produce collogen, thereby improving the appearance of aging skin, such as the lines known as “crow’s feet.”
