안녕하세요 보스턴 임박사입니다.
Elicio Therapeutics는 MIT의 Darrell Irvine 교수 연구실에서 2014년에 Nature에 발표한 “Amphiphile (AMP)” Platform을 기반으로 설립된 바이오텍 회사입니다.
Hitchhiking vaccines boost immunity – MIT News 2/16/2014
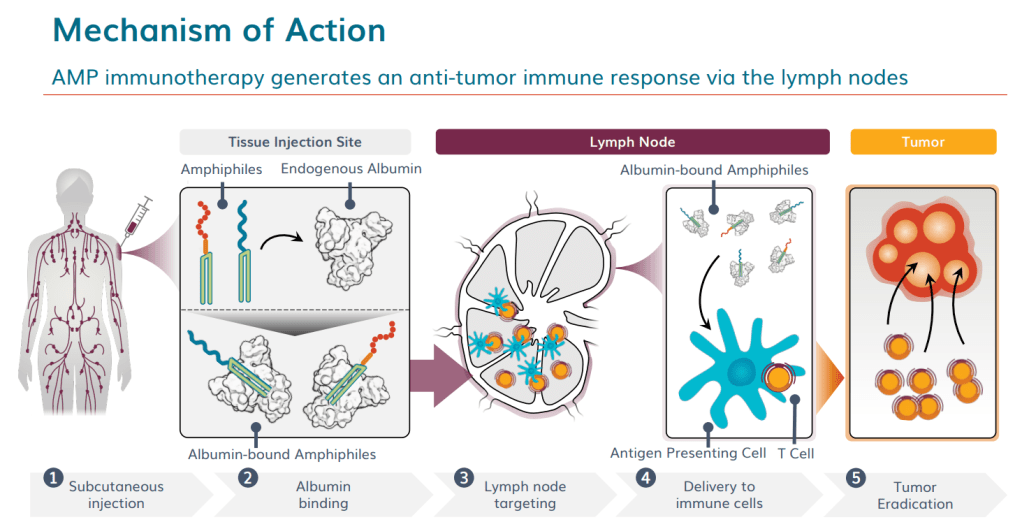
Now a team of engineers at MIT has developed a new way to deliver such vaccines directly to the lymph nodes, where huge populations of immune cells reside: These vaccines hitch a ride to the lymph nodes by latching on to the protein albumin, found in the bloodstream. In tests with mice, such vaccines produced very strong immune responses, the researchers report in the Feb. 16 online edition of Nature.
This approach could be especially useful for delivering HIV vaccines and for stimulating the body’s immune system to attack tumors, says Irvine, who is also a member of MIT’s Koch Institute for Integrative Cancer Research.
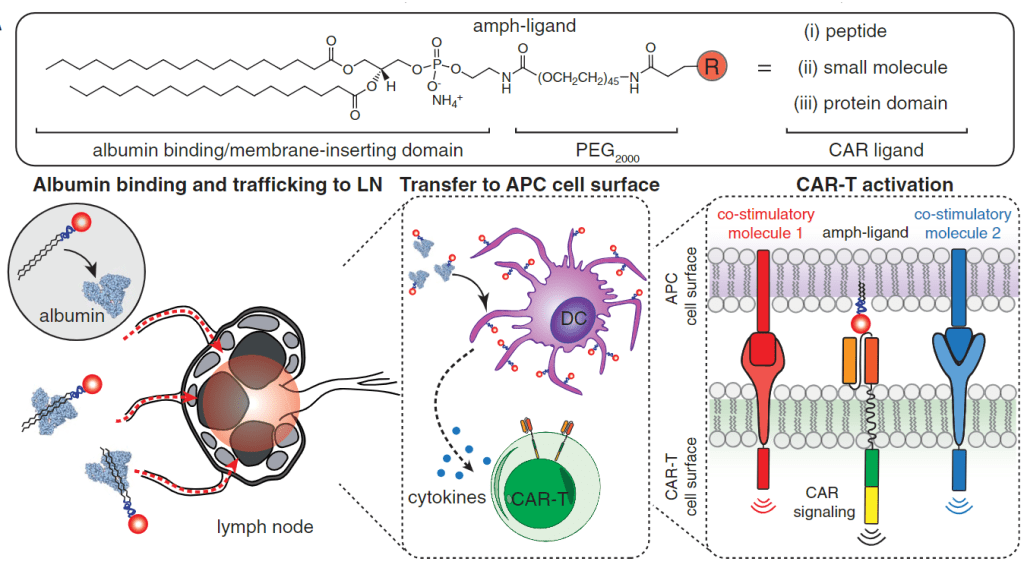
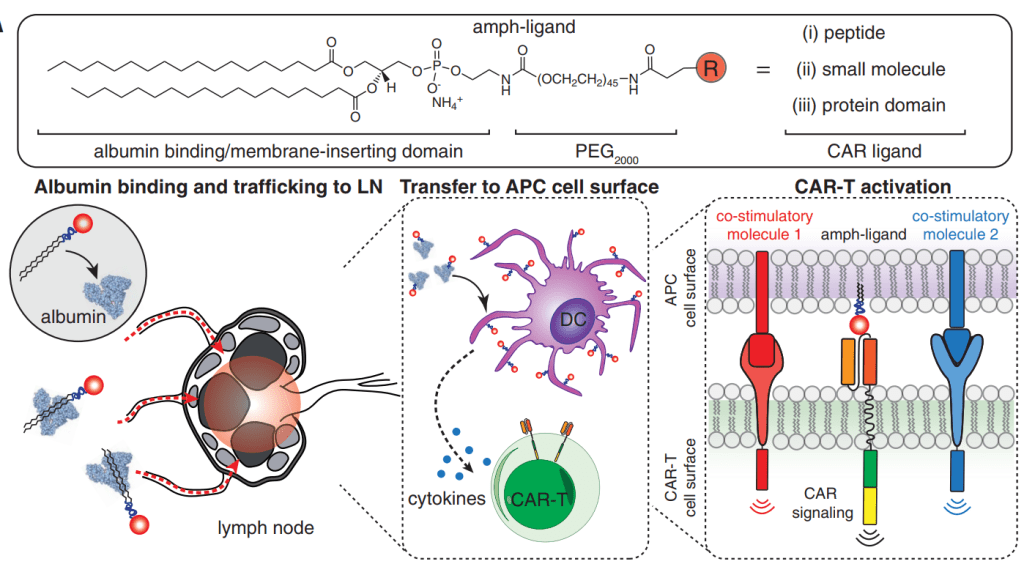
2019년에는 AMP platform을 이용해서 CAR-T cell therapy에 이용할 수 있슴을 보고하였습니다.
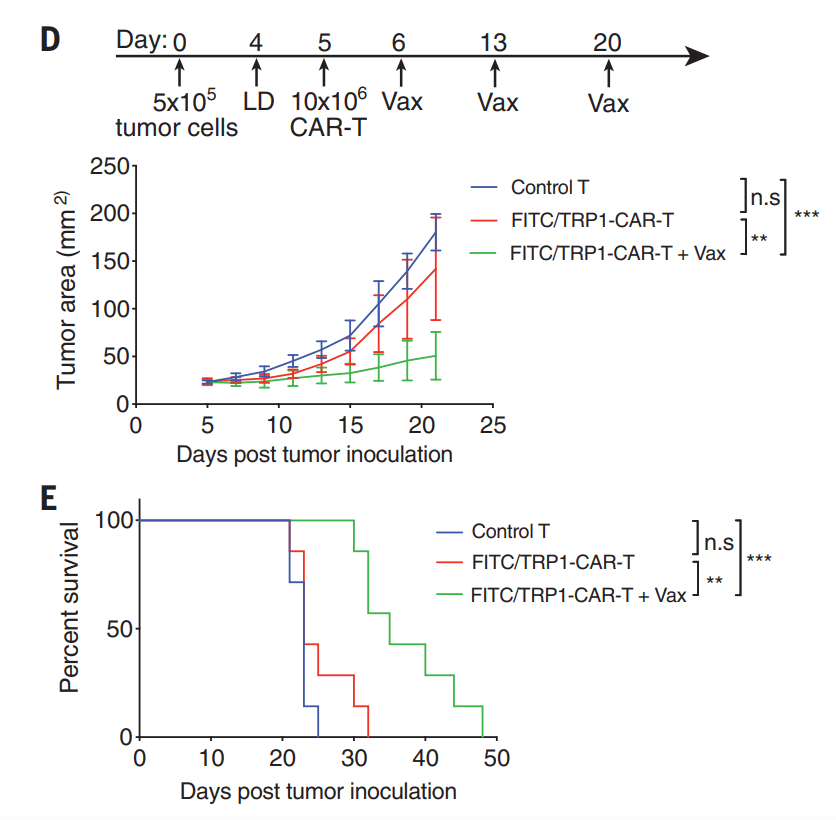
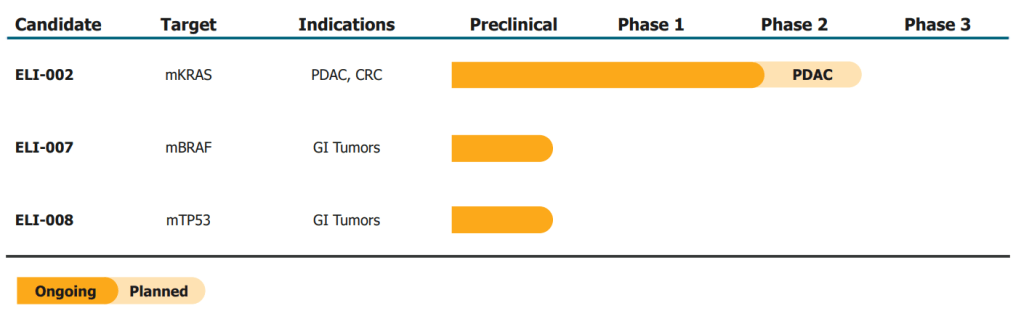
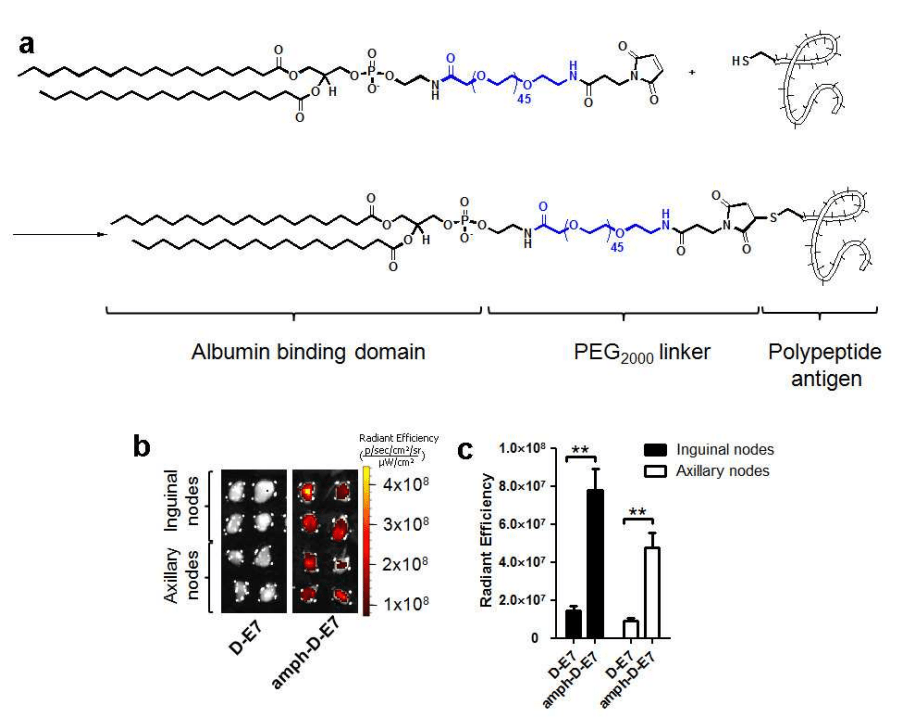
2021년에 $73 Million Series B를 하고 mKRAS cancer 대상한 ELI-002의 임상시험을 위해 노력하였습니다.
Elicio Therapeutics Secures a Total of $73 Million in Series B Financing – Business Wire 2/17/2021
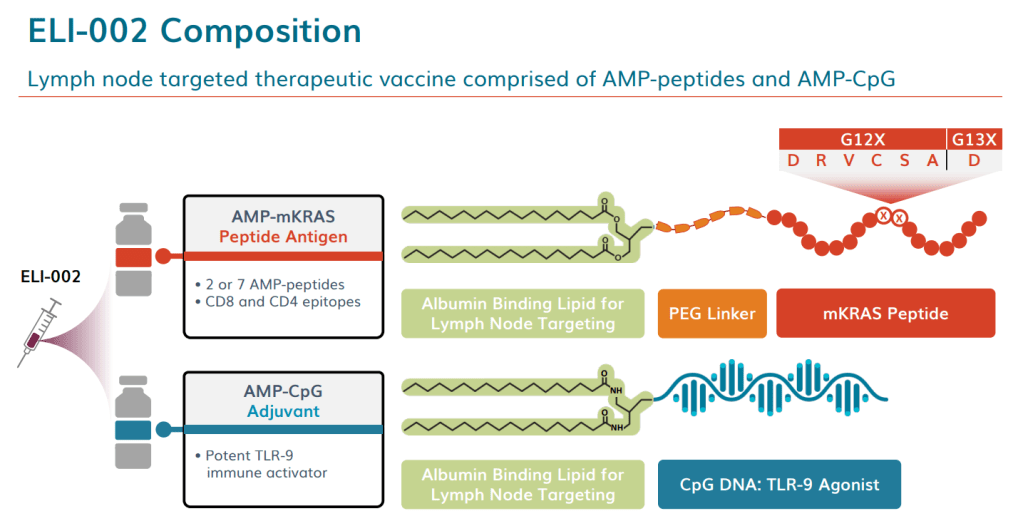
Proceeds will advance Elicio’s lead program ELI-002 into clinical trials in early 2021 for mutated KRAS (mKRAS) driven cancers, estimated to be 25% of all human solid tumors. ELI-002 contains two powerful components: The company’s proprietary immune stimulating Amphiphile (AMP)-CpG, an adjuvant, and AMP mKRAS peptides which target a broad spectrum of KRAS mutations that drive 97% of all KRAS-driven cancers.
1년반 후 IPO를 하기로 하려던 계획을 취소하고 대신 $40 Million Series C를 했습니다. 이 당시 ELI-002의 phase 1가 진행 중이었습니다.
Elicio Therapeutics has secured $37 million of its planned $40 million Series C, the company confirmed in an email to Endpoints. The Boston biotech had sought to list on Nasdaq as ELTX, with a draft registration statement filed in April 2021 and subsequent SEC filings showing ambitions to price between $12 to $14 apiece. The offering would’ve brought in about the same amount of capital as Elicio now seeks in its private round.
IPO 대신 6개월 후에 Angion Biomedica와 Reverse Merger에 의해 Nasdaq에 상장합니다.
Elicio Therapeutics Announces Completion of Merger with Angion Biomedica – Globe News Wire 6/1/2023
Elicio Therapeutics (Nasdaq: ELTX), a clinical-stage biotechnology company developing a pipeline of novel immunotherapies for the treatment of cancer, today announced the closing of its previously announced merger with Angion Biomedica Corp. The combined company will operate under the name Elicio Therapeutics, and its shares will commence trading on a 1-10 reverse split adjusted basis on June 2, 2023, on the Nasdaq Global Market under the ticker symbol “ELTX”.
Hitchhiking cancer vaccine makes progress in the clinic – MIT News 2/15/2024
By 2016, Irvine was ready to begin translating the vaccine from lab bench experiments to a patient-ready treatment, spinning out a new company, Elicio.
At Elicio, Irvine’s vaccine has evolved into a platform combining lipid-linked peptides with an immune adjuvant—no CAR T cells required. In 2021, the company began a clinical trial, AMPLIFY-201, of a vaccine named ELI-002, targeting cancers with mutations in the KRAS gene, with a focus on pancreatic ductal adenocarcinoma (PDAC). The company has initially tested a version that targets two, and Phase 1 and 2 studies of the version targeting all seven KRAS mutants are ongoing.
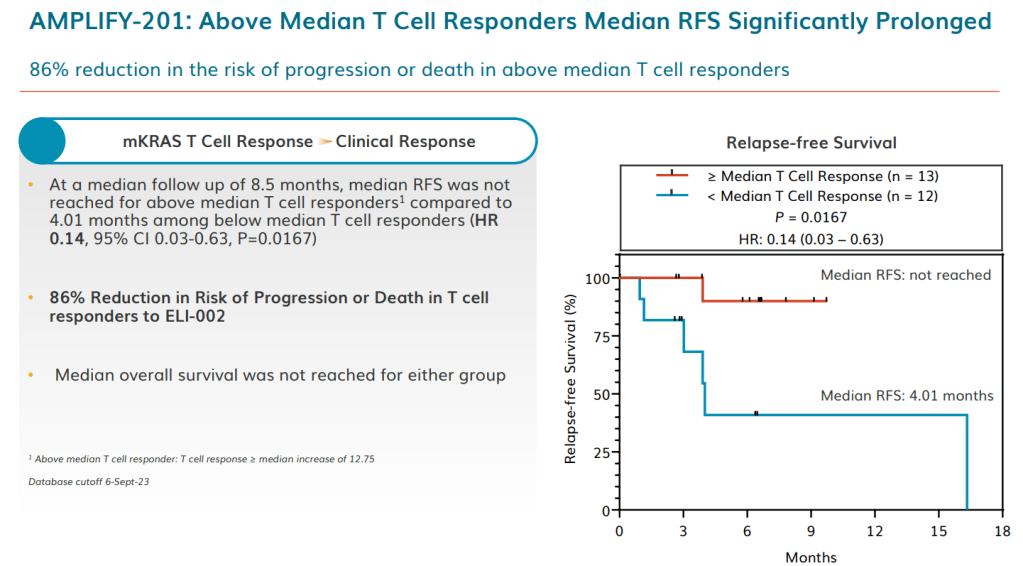
Data published last month in Nature Medicine from the Phase 1 clinical trial suggests that an effective therapeutic cancer vaccine could be on the horizon. The robust responses seen in the Irvine Lab’s mouse models have so far translated to the 25 patients (20 pancreatic, 5 colorectal) in the trial: 84% of patients showed an average 56-fold increase in the number of antitumor T cells, with complete elimination of blood biomarkers of residual tumor in 24%. Patients who had a strong immune response saw an 86% reduction in the risk of cancer progression or death. The vaccine was tolerated well by patients, with no serious side effects.
2024년에 발표한 Elicio Therapeutics의 Corporate Presentation은 아래와 같습니다.