(Picture: Mark C. McKenna, Founder & CEO of Mirador Therapeutics)
안녕하세요 보스턴 임박사입니다.
작년 6월에 Merck는 Prometheus Biosciences를 $10.8 Billion에 All-cash deal로 인수합니다. Merck의 Presentation을 보면 Prometheus의 Pipeline 중 PRA023 (MK-7240, Potential Best-In-Class & First-in-Class TL1A Antibody)에만 관심이 있습니다.
Prometheus에는 PRA023 말고도 PRA052 (CD30 ligand mAb)를 비롯한 다양한 Early-Stage Programs이 있었습니다. Merck는 이들 프로그램에는 관심이 없는 것 같습니다.

Merck Finalizes $10.8B Prometheus Buy – Biospace 6/19/2023
Merck finalized its acquisition of immune-focused Prometheus Biosciences for approximately $10.8 billion, scoring five clinical and pre-clinical candidates for inflammatory bowel and immune-mediated diseases on Friday.
Merck announced that Prometheus would now function as a fully-owned subsidiary. The leading candidate under Prometheus, originally designated for the treatment of ulcerative colitis and Crohn’s disease, has now been rebranded as MK-7240. This therapy serves as a catalyst, moving Merck further into the immunology space, an area in which the company has previously had limited involvement.
Original story published April 17:
Merck Acquires Prometheus Biosciences for Nearly $11 Billion
As per the terms of the all-cash agreement, Merck will buy all of Prometheus’ outstanding shares for $200 a piece, representing a 75% premium to Prometheus’ Friday closing price of $114. The companies expect to close the deal in the third quarter of 2023.
At the center of the transaction is Prometheus’ investigational monoclonal antibody, PRA023, which is set to enter late-stage studies later this year for ulcerative colitis (UC) and Crohn’s disease (CD). The drug targets and inhibits the action of tumor necrosis factor (TNF)-like ligand 1A (TL1A), a key immune factor that plays a role in both inflammation and fibrosis in intestinal immune-mediated disease.
In December, Prometheus announced results from Phase II and IIa studies that showed promising efficacy and safety in both inflammatory bowel diseases.
In the Phase II ARTEMS-UC study, 26.5% of patients with moderately-to-severely active UC reached clinical remission 12 weeks after receiving PRA023, as opposed to 1.5% of placebo comparators. Prometheus’ candidate also demonstrated superior endoscopic improvement and met all of the trial’s secondary endpoints.
PRA023 also performed well in the Phase IIa APOLLO-CD study, eliciting a 49.1% clinical remission rate in patients with moderately-to-severely active disease, compared with a 16% historical placebo rate. The candidate also led to higher endoscopic response and improved several markers of fibrosis and inflammation.
Both trials enrolled patients who had failed prior lines of therapy.
Prometheus is also evaluating PRA023 in systemic sclerosis-associated interstitial lung disease in Phase II assessments. In March 2022, the company kicked off the Phase II ATHENA-SSc-ILD study in this indication, with topline results expected in the first half of 2024.
Aside from PRA023, Prometheus is advancing four other candidates for immune-mediated indications, all of which will join Merck’s fold after the buyout.
Prometheus’ stock soared 70% in pre-market trading Monday, while Merck’s fell 1%.
Merck의 Prometheus Biosciences 인수가 마무리되고 나서 Mark C. McKenna를 비롯한 ex-Prometheus Biosciences Team은 다시 Mirador Therapeutics를 설립합니다. Arch Ventures, Orbimed와 같은 좋은 투자자들이 든든히 뒤를 받쳐준 덥분에 $100 Million Series A를 하려던 계획은 실제로 4배 규모의 시리즈 A로 마무리짓게 됩니다.
After being bought out by Merck in June 2023 for nearly $11 billion, some of the ex-brass from Prometheus Biosciences headed to a spear-shaped peninsula in Mexico surrounded on three sides by white sand beaches, Pacific Ocean waters, and lush tropical flora for some rest and relaxation. But playing golf and taking in the sea breeze in the coveted hideaway of Punta Minta wasn’t enough to keep Mark C. McKenna, the former chairman, president, and CEO of Prometheus Biosciences, from getting back into the game of making medicines.
“The transaction with Merck was bittersweet,” McKenna told GEN Edge. “They were the right buyers, and it was the right time to do it… But there was a lot of unfinished business at Prometheus. There was a realization that we could all go work for different companies, or we could come back together and finish the mission. The field of data science is evolving so rapidly, so we were only scratching the surface.”
About three-quarters of a year after the buy-out, McKenna, with some Prometheus veterans and new additions, unveiled Mirador Therapeutics with the vision to bring “end-to-end” precision medicine into immunology and inflammation. Behind a $400 million Series A and their proprietary platform, Mirador360, Mirador plans to leverage open-source human genetics data and cutting-edge data science, which, according to McKenna, will enable the company to file for investigational new drugs (INDs) by 2025.
“We believe that based on our experience in this category, we have a unique vantage point,” said McKenna, alluding to the company’s name, which directly translates to viewpoint in Spanish.
Prometheus veterans Olivier Laurent, PhD, Allison Luo, MD, Tim Andrews, Vika Brough, Nori Ebersole, and Jordan Zwick will join McKenna at Mirador Therapeutics in San Diego, CA. As for new faces, Mirador has brought on William J. Sandborn, MD, who previously served as a professor of medicine and chief of the division of gastroenterology at the University of California, San Diego, for ten years and co-founded Santarus and Shoreline Biosciences.
작년 3월에 발표된 Prometheus Biosciencs 팀 Executive Team 8 명 중 4명이 Mirador에 조인을 합니다.

그리고 UCSD의 William J. Sanborn 박사가 조인을 하기로 되어 있습니다. 그는 IBD 등 Gastroenterology MD 입니다.

“It’s what you do with the data.”
For Prometheus, that surface consisted of a clinical database and associated biobank exclusively licensed from Cedars-Sinai Medical Center. It included more than 200,000 samples linked to extensive clinical data from 20,000+ patients collected over 20 years. According to McKenna, the problem with that is that technology changes.
Prometheus360이라고 불리던 지난 20년간 모은 2만개 이상의 환자 시료를 포함한 20만개 이상의 샘플이 있습니다.
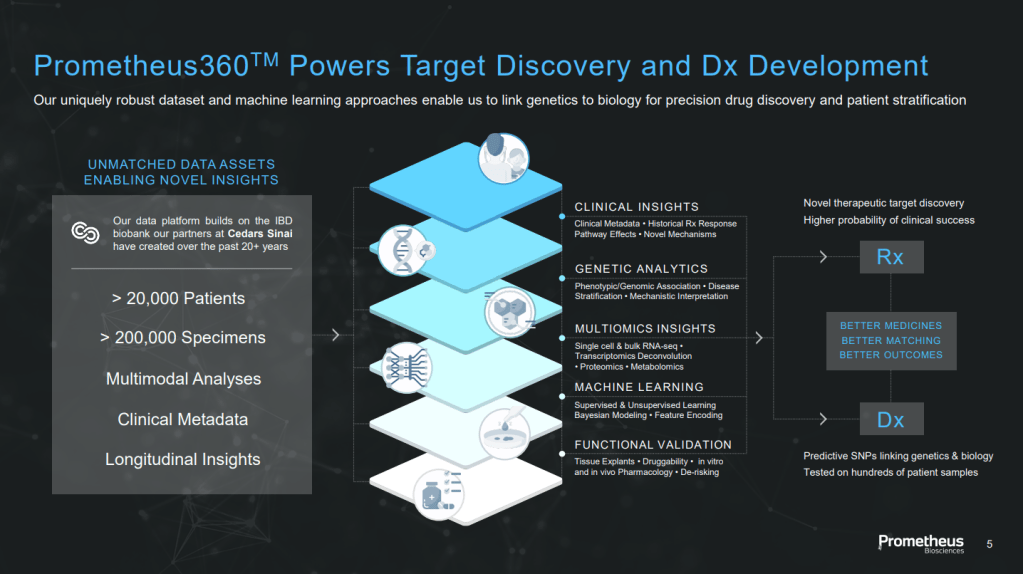
이것들을 이용한 Data Science와 Genetics를 통해 새로운 표적을 발굴하는 것이 Prometheus Biosciences의 Platform Engine이었습니다. Patients Samples로 Inflammatory Diseases의 Genetics를 해독하고 새로운 표적을 발굴했는데 이런 과정에서 발굴된 것 중 하나가 PRA023 (MK-7240) 이었습니다. 검증된 플랫폼인 것이죠. McKenna는 소화기 (Gastrointestinal), 폐 및 피부 질환에 관심이 많다고 말했습니다. 이를 위해 학계와 파트너쉽도 하고 좋은 Asset이 있으면 Licensing-In 하려는 계획도 밝혔습니다.
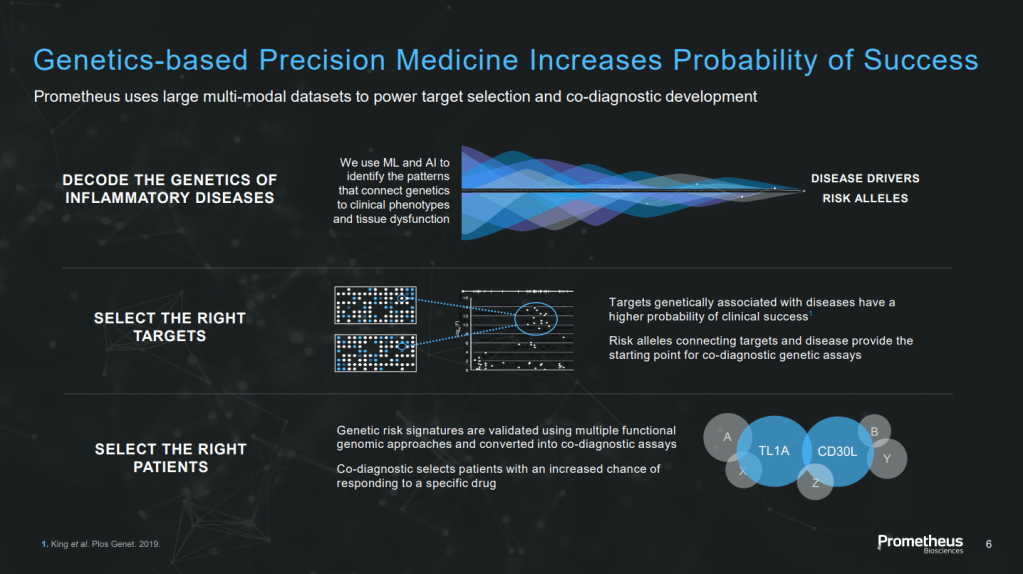
McKenna said, “One of the things that big pharma, whether Regeneron or others, is realizing is that sequencing techniques and data science are evolving so quickly, and we need them to continue to invest in democratizing data and sequencing patients. Thank you, George [Yancopoulos] from Regeneron, for all you do at the UK Biobank and other places because that allows for innovation. I don’t believe the data itself is what’s proprietary. I think that what you do with the data is. The technologies allowing these data sets to be interwoven and translated more rapidly than ever weren’t available months ago.”
Mirador’s scope of indications will go much broader than just inflammatory bowel disease. McKenna said Mirador is building a data cohort that straddles the disease areas they’re interested in—gastrointestinal tract, lung, and skin diseases—from a combination of open-source data and proprietary academic partnerships.
End-to-end precision medicine
According to McKenna, end-to-end means Mirador will discover and validate genetic associations with immuno-fibrotic diseases, identify novel therapeutic targets and optimal target-target pairs for potential combination therapies, develop diagnostics, and stratify heterogeneous patient populations for precise clinical development.
“No one’s been able to do this before,” said McKenna. “We can put this on steroids and move the field further. It’s not the beginning but certainly not the end either.”
In the last twenty years, McKenna said that he’s learned several lessons, none more important than properly managing and stratifying risk. This is particularly applicable to committing to using a single therapeutic modality. McKenna said that Mirador will not be afraid to try novel approaches from a modality perspective, but it will not be at the core of what they do.
“If you think about the evolution here… [In the early days, you started] with an entrepreneur or scientist with one idea and a biased binary outcome,” said McKenna. “Then we shifted to this platform-focused idea: create all this data and then ask, is there actually a product? In version 3.0, it is more like a pipeline and a drug. We are focused on and have expertise in biologics. But we’re agnostic to what it is once it’s in the clinic, and we will have approaches with biologics working on targets that we find interesting and can be best-in-class. You have got to think about this for stratification very carefully.”
Momentum-gaining investments
The massive $400 million round of funding began through discussions with six investors, some of whom had previously worked with Prometheus; some were new relationships.
“I wanted to work with people who had a big vision for doing transformational stuff and had a track record doing so, and I’m not sure you could have found better leads than ARCH [Venture Partners], OrbiMed, and Fairmount [Partners],” said McKenna. “Each of them brings different things to the table. At ARCH, [co-founder and managing director] Bob Nelson has big visionary ideas. [Managing director] Kristina Burrow understands how companies operate. You have OrbiMed’s public-market understanding. You have Fairmount and their ability to build companies. Then you have these different investors, like Fidelity—when was the last time Fidelity entered a Series A? So, we hand-selected these investors.”
McKenna said the $400 million will go to multiple programs, INDs, and clinical data. It will allow Mirador to in-license external innovation that would benefit from our platform.
“We never went out and said, We want to raise $400 million,” said McKenna. “We said we will raise [$100 million], which started as a snowball. This is more dilution than necessary in the early days. We want to have a war chest to go in, create value, and build a pursuing medicine engine that people just haven’t been able to do.”
McKenna also said that Mirador will engage in external innovation, finding programs and companies that benefit from its platform, expertise, and know-how to accelerate the company’s pace.
“Time is the new currency, particularly in biotech, and you see a lot of companies spending years and years to figure it out and burning a lot of cash to get there,” said McKenna. “One of the most remarkable things about the journey from Prometheus was that, in three and a half years, we created value, and the goal was never to build something to sell—the goal was to build a great company. If we build a great company and focus on inputs, the output will come. That’s exactly what we’re doing here [at Mirador]. This team is fired up, and it’s not about the money; it’s about what kind of impact we can actually have.”
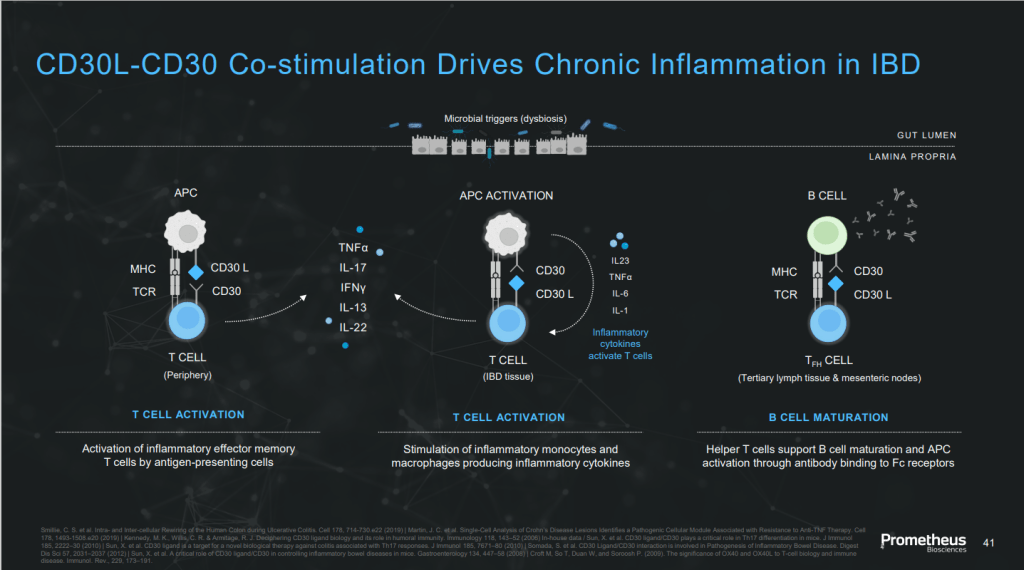
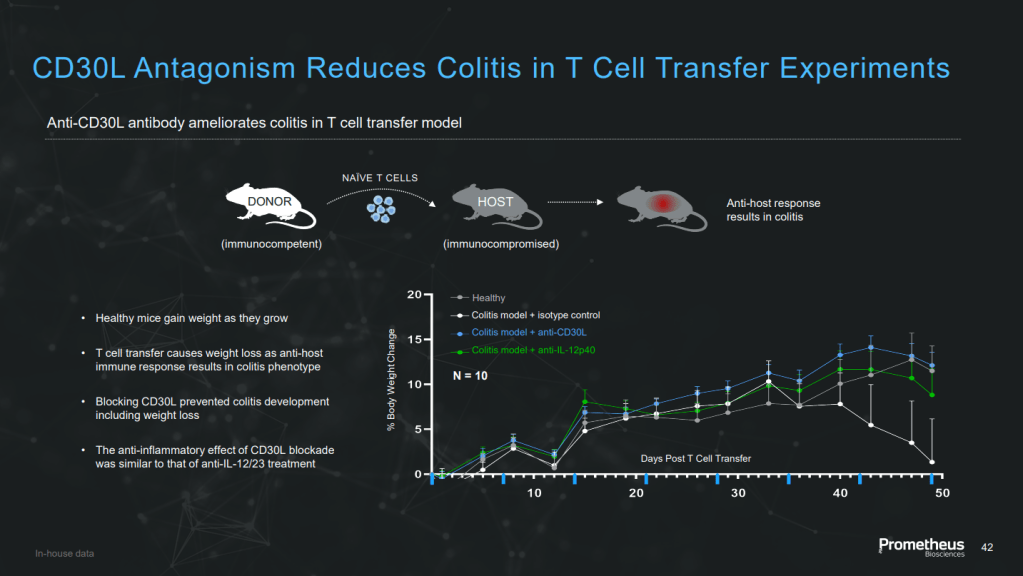
Merck와의 딜로 인수는 되었지만 Merck가 관심이 없는 임상1상 중인 약물 PRA052 (CD30 ligand mAb)이 있습니다. 최근 발표 자료에 의하면 만성 IBD 질환에 드는 약물입니다. 이 약물을 Merck로 부터 인수할지 여부도 궁금하고 향후 Mirador가 어떤 행보를 할지 궁금합니다.