안녕하세요 보스턴 임박사입니다.
Entos Pharmaceuticals는 Cananda의 Dalhousie University에 있는 Roy Duncan 교수 연구실의 Fusogenic Proteolipid Vehicle (PLV) Delivery Platform을 기반으로 한 Gene Therapy Company입니다.

Roy Duncan교수의 Fusogenic PLV Platform은 Roy Duncan 교수가 세운 Fusogenix로 IP가 이전되었다가 Innovascreen으로 합병되어 2010년대에 기술 개발이 이루어집니다. Roy Duncan 교수는 2000년에 Embo Journal에 FAST Protein에 대해 최초로 보고합니다.
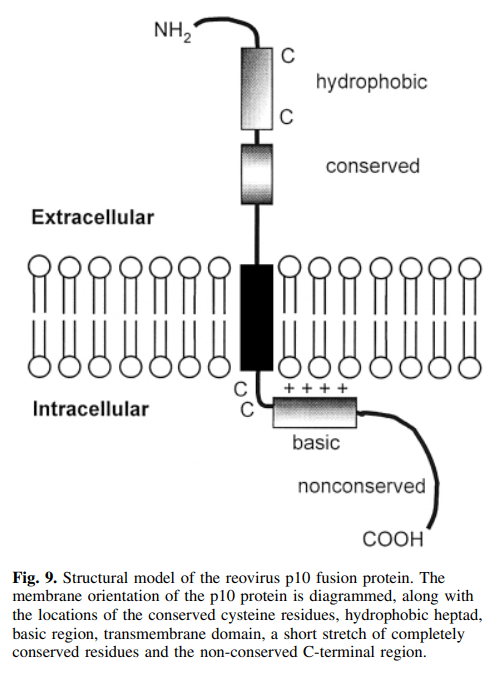
Roy Duncan 교수는 2010년부터 Fusogenic Reovirus를 이용하여 Fusion-Associated Small Transmembrane (FAST) Protein을 개발하였는데 이에 대해 본인이 2019년에 Annual Reviews에 기고한 논문이 있습니다.
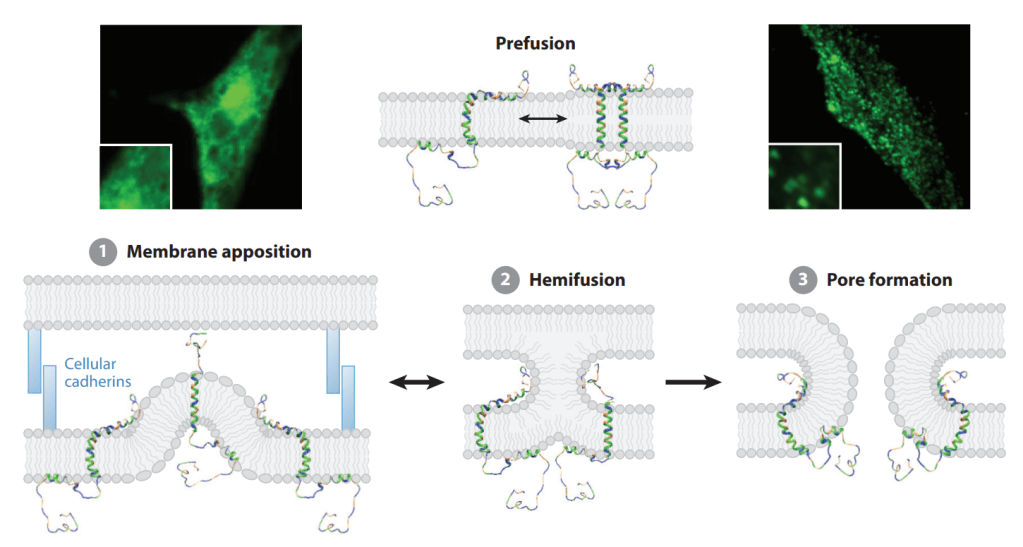
Entos Pharmaceuticals Founder이자 CEO인 John Lewis 박사는 학회발표나 논문기고를 통해 Fusogenix FAST를 알리는데 열심히 노력을 했습니다.
COVID-19 Pandemic 기간 동안에 Entos Pharmaceuticals는 Covigenix DNA vaccine VAX-001의 임상을 진행하였습니다.
Aegis and Entos commence dosing in trial of Covid-19 DNA vaccine – Clinical Triasl Arena 4/19/2021
US company Aegis Life has initiated dosing of the first participants in a Phase I/II clinical trial with parent company Entos Pharmaceuticals’ novel DNA Covid-19 vaccine, Covigenix VAX-001, to induce immunity against SARS-CoV-2.
Entos Pharmaceuticals has obtained approval from the South African Health Products Regulatory Authority (SAHPRA) to commence a Phase II clinical trial of its Covid-19 vaccine candidate, Covigenix VAX-001, in the country.
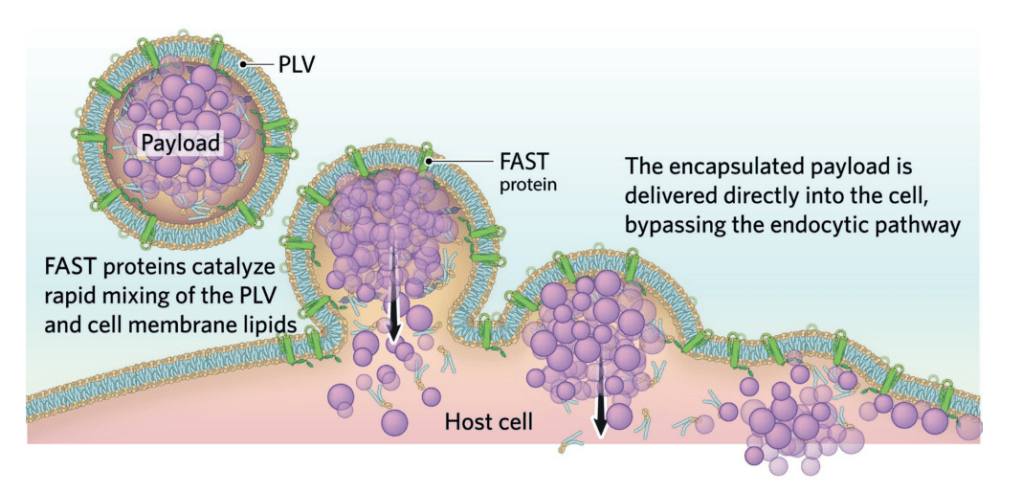
Made using the Entos’ Fusogenix proteolipid vehicle (PLV) nucleic acid delivery platform, the deoxyribonucleic acid (DNA) vaccine encodes the SARS-CoV-2 Spike protein. It also includes two genetic adjuvants to induce the innate and adaptive immune systems, offering efficient and long-term protection from Covid-19.
The Fusogenix platform leverages a new mechanism of action to carry molecules directly into the cytosol of intended cells in an intact and unaltered manner.
The latest approval is based on positive Phase I data, which showed that Covigenix VAX-001 met all the safety goals without any serious adverse events noted in the trial.
이런 노력의 결과로 최근 BioMarin과 Eli Lilly와 잇달아 연구계약을 맺게 됩니다. 특히 Eli Lilly는 $50 Million Upfront 포함 $400 Million에 exclusive right을 얻게 되어 Eli Lilly가 개발 중인 Nucleic Acid Therapy에 Fusogenix PLV Platform을 사용할 계획입니다.
“As a company committed to addressing the unmet therapeutic needs of patients living with genetic diseases, BioMarin values novel technologies that enable the development of transformative therapies,” said Brinda Balakrishnan, M.D., Ph.D., Group Vice President, Corporate and Business Development at BioMarin. “We believe that Entos’ Fusogenix platform offers potentially unique benefits for safe and effective tissue targeting compared with other lipid-based delivery systems. The Fusogenix PLV formulations generated under this agreement are a critical first step in determining how we may incorporate this promising platform into our drug development efforts.”
Eli Lilly taps Entos’ delivery tech in $50M-plus nucleic acid pact – Fierce Biotech 1/6/2022
Entos Pharmaceuticals’ star is rising. Just a few short months after the biotech’s nucleic acid delivery tech helped land a deal with BioMarin, the Edmonton, Alberta-based Canadian company has teamed up with Eli Lilly for $50 million upfront to drive its proteolipid vehicles (PLVs) toward a range of nervous system targets.
Eli Lilly has picked up exclusive right to Entos’ Fusogenix nucleic acid delivery technology, which it will use to research, develop and potentially sell nucleic acid-based therapeutics against targets in the central and peripheral nervous systems, the companies said Thursday.
The deal breaks down like this: Lilly and Entos will collaborate on multiple programs that wed Lilly-supplied therapeutic cargo to Entos’ PLVs. Entos is on deck to generate, develop and optimize those PLVs on its proprietary Fusogenix platform. Lilly will then select PLVs to take into the clinic.
Entos is in line to receive $50 million upfront from the R&D pact, which includes an equity investment in the company by Lilly. Each program under the collaboration could net Entos up to $400 million more in potential development and commercial milestones, plus royalties should any of the products reach the market.
Entos’ PLVs are formulated with so-called fusion-associated small transmembrane proteins, or FAST proteins, plus neutral lipids for better tolerability. Fusogenix, which can be used to deliver a range of therapy types, such as gene therapy, mRNA, miRNA, RNAi and CRISPR, delivers mRNA or DNA into target cells through direct fusion.
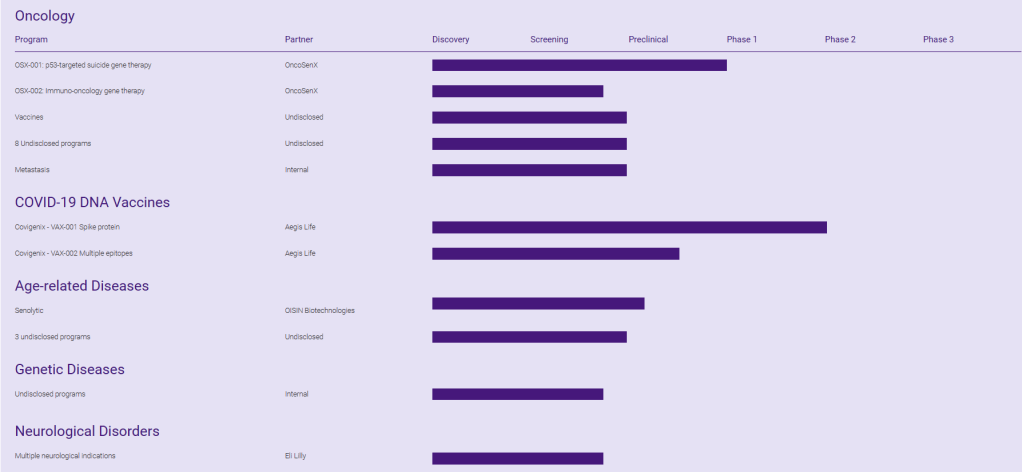
Entos Pharmaceuticals의 Pipeline은 아래와 같습니다. Fusogenix PLV가 Virus Vector나 LNP를 대체할 차세대 Gene Delivery System으로 상용화가 가능할지 주목됩니다.