
안녕하세요 보스턴 임박사입니다.
Canada Toronto의 Princess Margaret Hospital의 Prostate Cancer Prevention Center의 대표였던 Neil Fleshner 박사는 2017년에 자신의 환자의 Radioligand 치료를 위해 독일로 보내야 하는 경험을 하게 되면서 Point Biopharma를 설립하고 경영진을 모으게 됩니다. Joe McCann박사를 CEO로, Neil Fleshner 박사는 CMO를 그리고 Michael Gottlieb는 CFO로 해서 Radioisotope supply와 생산기술 등을 전략적 제휴를 통해서 만들어내게 됩니다.
Point was founded after Neil Fleshner, the Love Chair in Prostate Cancer Prevention at Toronto’s Princess Margaret Hospital, had to send a patient to Germany in 2017 to receive radioligand treatment. He sought to make such therapy more accessible, bringing together a corporate team.
Point Biopharma Launches With Radiopharmaceutical Development Focus – Biospace 2/25/2020
POINT Biopharma, a newly formed pharmaceutical company, is combining a seasoned management team with strategic partnerships in radio-isotope supply, manufacturing technology and novel direct to patient targeting to revolutionize radiopharmaceutical drug development and commercialization. Working closely with its scientific advisors, the Company anticipates commencement of its clinical trial programs in 2020.
Dr. Joe McCann, PhD has assumed the role of Chief Executive Officer. An industry veteran with more than 10 years of proven radiopharmaceutical experience, Joe was most recently the CEO of Centre for Probe Development and Commercialization. Dr. Neil Fleshner, an uro-oncologist, with more than 400 authored papers, has assumed the role of Chief Medical Officer and Michael Gottlieb, CPA, the former head of Sanofi Genzyme Canada’s Rare Disease Business joins as Chief Financial and Commercial Officer.
177Lu-PSMA Precision Radiopharmaceuticals인 PNT2002의 임상3상을 시작하면서 $20 Million Series A를 하게 됩니다.
“So far 2020 has been a very productive year for POINT,” Dr. McCann continued, “including the announcement of the Phase 3 clinical trial for PNT2002, our 177Lu-PSMA radiotherapeutic for the treatment of metastatic castrate-resistant prostate cancer, as well as the purchase and build-out of our 77,000 sqft radioligand manufacturing facility in Indianapolis, Indiana. Now, with the successful close of our Series A financing, POINT is well positioned to execute on our mission of making radioligands applicable to more cancers, accessible to more people, thereby improving the lives of patients and their families.”
그리고 7개월 후에 SPAC 상장에 의해 NASDAQ에 등록되고 이 때 $465 Million의 자금을 확보하게 됩니다. 이 자금으로 PNT2002의 임상3상과 Manufacturing Facility를 Indianapolis에 세우기 위한 충분한 자금이 확보된 것입니다.
POINT Biopharma Inc. (“POINT”), a late-stage biopharmaceutical company dedicated to bringing the many benefits of precision radiopharmaceutical therapies to patients with cancer, and Therapeutics Acquisition Corp., d/b/a Research Alliance Corp. I (Nasdaq: RACA) (“RACA”), a special purpose acquisition company, or SPAC, sponsored by RA Capital Management, today announced they have entered into a definitive business combination agreement
A group of top-tier investors has committed to participate in the transaction through a common stock PIPE of approximately $165 million at $10.00 per share. Investors in the PIPE include lead investor RA Capital Management, an affiliate of RACA’s sponsor, as well as Johnson & Johnson Innovation – JJDC, Inc., Surveyor Capital (a Citadel company), Farallon Capital Management, L.L.C., BVF Partners L.P., Boxer Capital, Sphera Healthcare, Woodline Partners LP, Suvretta Capital, Fairmount Funds, and Perceptive Advisors. Assuming no redemptions are exercised, the Combined Company is expected to receive net proceeds of approximately $300 million at the closing of the transaction (inclusive of the trust account balance and the proceeds from the PIPE).
With the funds raised from this transaction, POINT will be well financed to complete our two Phase 3 trials for radioligands to treat prostate and neuroendocrine cancers, advance our early-stage pipeline, and complete construction on our manufacturing facility in Indianapolis, Indiana.
SPAC 상장 1년여 후 Lantheus와 $260 Million upfront를 포함해서 $1.8 Billion까지 계약을 하고 PNT2002의 상용화 시 20% royalty, PNT2003의 경우는 15% rotyalty를 받을 수 있는 딜을 하게 됩니다.
Lantheus is paying $260 million upfront for a double bill of licenses for two of POINT Biopharma’s radiopharmaceutical oncology candidates, with another $1.8 billion tied up in biobucks.
Under the agreements, POINT will continue to fund and complete its phase 3 SPLASH trial for PNT2002, a prostate-specific membrane antigen (PSMA)-targeting 177Lu-based radiopharmaceutical therapy for metastatic castration-resistant prostate cancer. After that, Lantheus will work with POINT to file the therapy for FDA approval.
The other candidate is PNT2003, a somatostatin receptor-targeted radioligand in development for gastroenteropancreatic neuroendocrine tumors. POINT will facilitate completion of the ongoing University Health Network-sponsored study in Canada, while Lantheus will prepare and submit the regulatory filings in the U.S.
Should the two assets secure FDA approval and then hit commercial milestones, POINT could be in line for up to $1.8 billion in biobucks plus royalties of 20% and 15% for PNT2002 and PNT2003, respectively.
그리고 1년 후에는 $125 Million의 유상증자를 하게 됩니다. PNT2002의 pivotal clinical trial 및 NDA Filing에 필요한 충분한 자금이 확보되었다고 할 수 있습니다.
POINT Biopharma Prices Public Offering of Common Stock – Globe News Wire 9/13/2022
The gross proceeds to the Company from the offering, before deducting underwriting discounts and commissions and other estimated offering expenses, are expected to be approximately $125 million.
2023년에 Eli Lilly는 준비된 Point Biopharma를 인수하기로 결정하게 됩니다. $1.4 Billion의 시총입니다. 전일 종가에 비해 87% 프리미엄의 좋은 딜이었습니다.
POINT’s lead programs are in late-phase development. PNT20021 is a prostate-specific membrane antigen (PSMA) targeted radioligand therapy in development for patients with metastatic castration-resistant prostate cancer (mCRPC) after progression on hormonal treatment. Topline data from this study are expected in the fourth quarter of 2023. PNT20031 is a somatostatin receptor (SSTR) targeted radioligand therapy in development for the treatment of patients with gastroenteropancreatic neuroendocrine tumors (GEP-NETs). Beyond the late-stage clinical pipeline, POINT has several additional programs in earlier stages of clinical and preclinical development. Additionally, POINT operates a 180,000-square-foot radiopharmaceutical manufacturing campus in Indianapolis, as well as a radiopharmaceutical research and development center in Toronto. These facilities will be utilized alongside POINT’s extensive network of supply chain partners for sourcing radioisotopes and their precursors.
Lilly will commence a tender offer to acquire all outstanding shares of POINT for a purchase price of $12.50 per share in cash (an aggregate of approximately $1.4 billion) payable at closing. The transaction has been approved by the boards of directors of both companies. The purchase price payable at closing represents a premium of approximately 87% to POINT’s closing stock price on Oct. 2, 2023, the last trading day before the announcement of the transaction, and 68% to the 30-day volume-weighted average price. POINT’s board of directors unanimously recommends that POINT’s stockholders tender their shares in the tender offer.
현재의 파이프라인과 2023년 6월 Investor Day Presentation을 아래에 올립니다. PNT2002와 PNT2003은 임상3상에 있꼬 PNT2004는 임상1상을 진행 중에 있습니다.
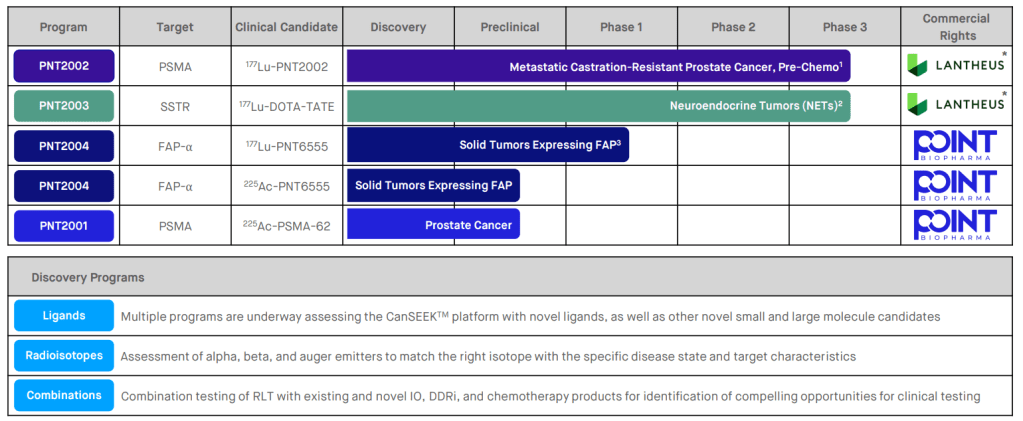
PNT2002는 Novartis의 Pluvicto의 경쟁제품이 될 것인데 승인이 되어서 전립선암 환자들에게 도움이 되기를 바랍니다.
BIOTECH (104) Novartis: Pluvicto (177Lu-PSMA-617) – the First Precision Radiopharmaceuticals
