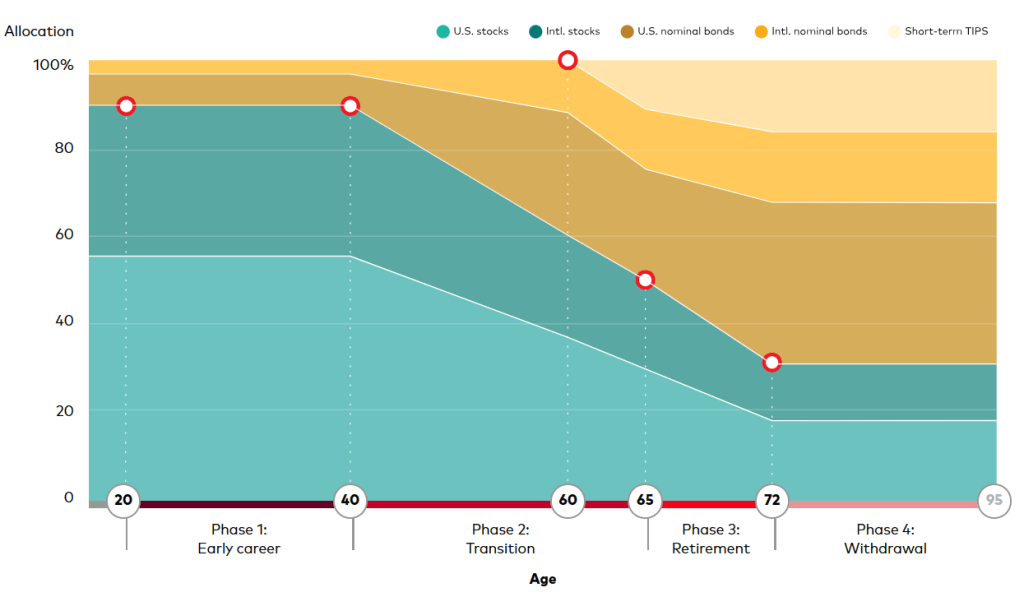
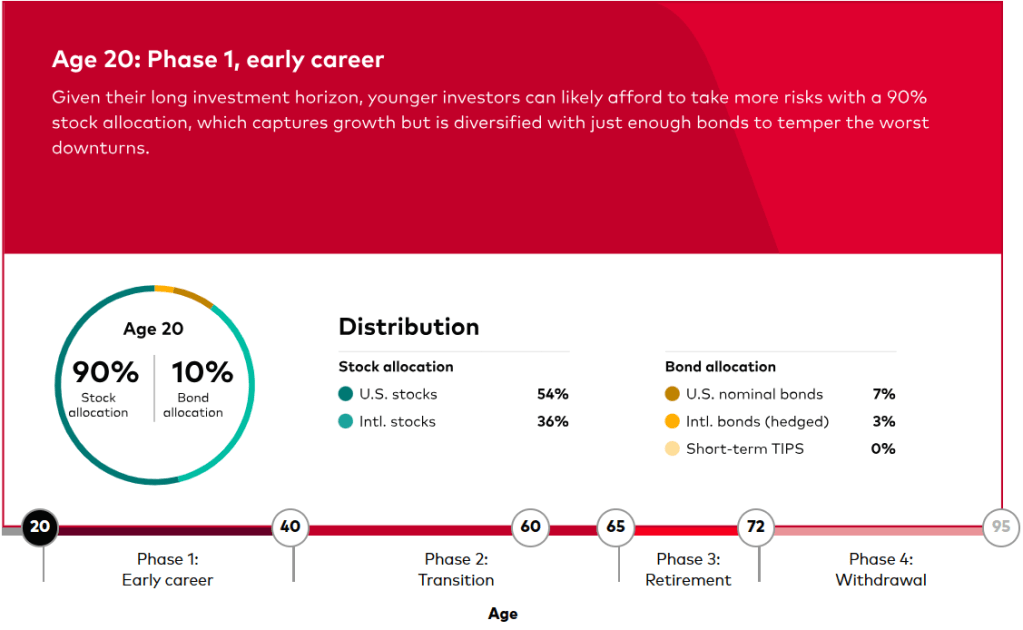
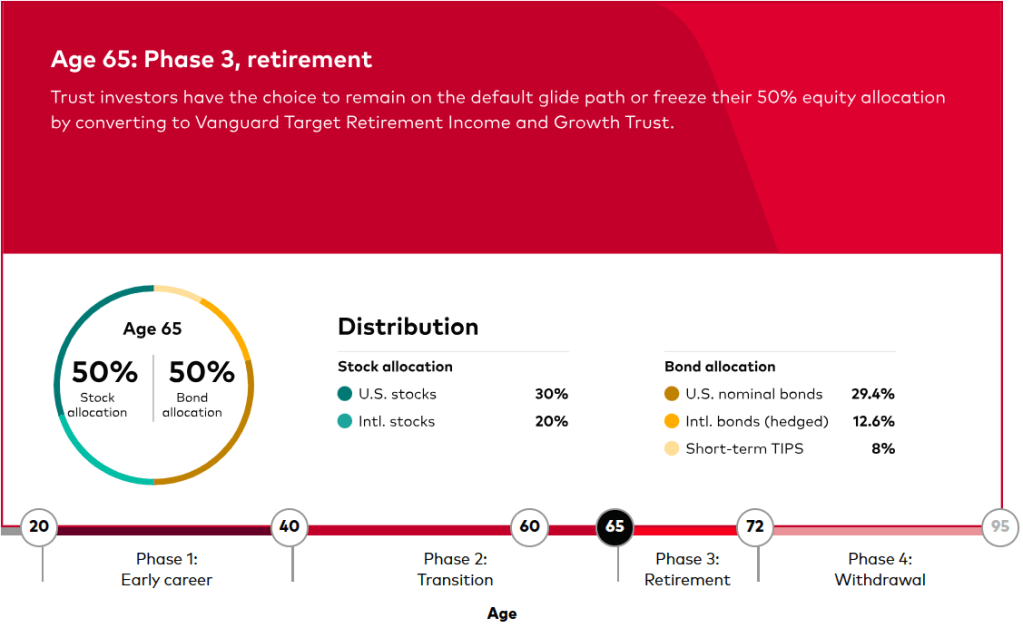
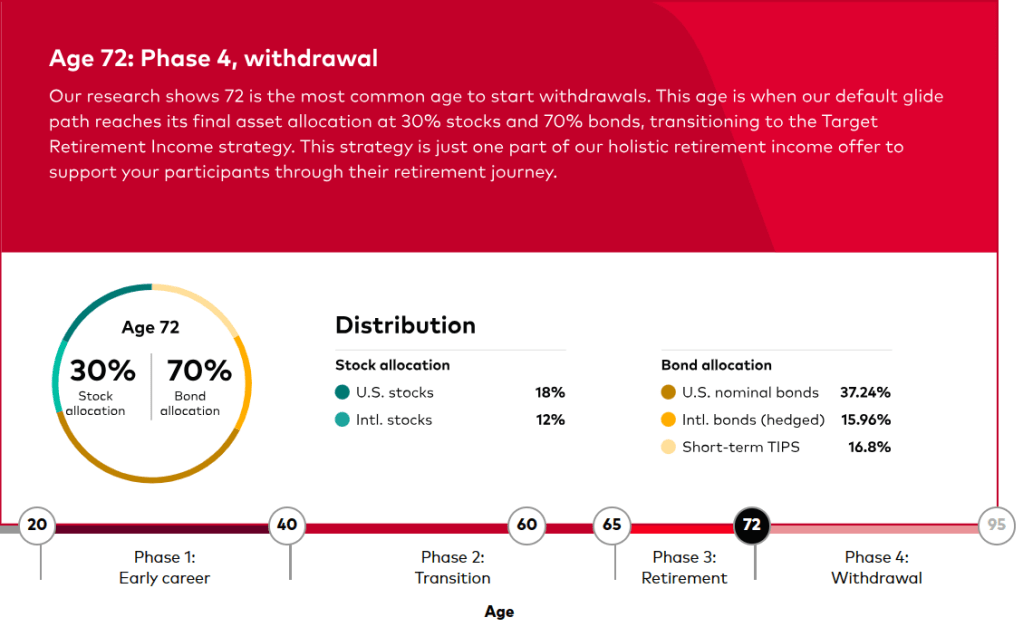
(Picture: Ugur Sahin, PhD, Co-Founder & CEO and Özlem Türeci, PhD, Co-Founder & CMO, BioNTech SE)
BioNTech Acquires Neon Therapeutics to Bolster Immuno-Oncology Pipeline – Biospace 1/17/2020
Mainz, Germany-based BioNTech announced it was acquiring Cambridge, Massachusetts-based Neon Therapeutics in an all-stock deal valued at about $67 million.
Neon’s focus is on neoantigen therapies that have the potential to be both vaccines and T-cell therapeutics. Neoantigens are immune targets generated by mutations inherent in tumors. Its most advanced program is NEO-PTC-01, a personalized neoantigen-targeted T-cell therapy. It is derived of multiple T-cell populations that target the most relevant neoantigens from each patient’s cancer. The company is also working on a precision T-cell therapy program that targets shared neoantigens in specific, genetically defined patient populations. The lead program from that endeavor is NEO-STC-01, which targets shared RAS neoantigens.
Neon Therapeutics was founded in 2015 by Third Rock Ventures. The company was one of BioSpace’s NextGen Class of 2017 biotech startups to watch.
“This acquisition fits with our strategy to expand our capabilities and build our presence in the U.S. and further strengthens our immunotherapy pipeline,” said Ugur Sahin, co-founder and chief executive officer of BioNTech. “I am particularly excited about the adoptive T-cell and neoantigen TCR therapies being developed by Neon, which are complementary to our pipeline and our focus on solid tumors.”
Under the terms of the deal, after the acquisition Neon will merge with Endor Lights, a wholly-owned subsidiary of BioNTech incorporated in Delaware. It will then become a wholly-owned subsidiary of BioNTech. At the close of the deal, BioNTech will issue 0.063 American Depositary Shares (ADS) to Neon shareholders in exchange for each of their Neon shares. This exchange ratio implies a deal value of $67 million, or $2.19 per Neon share, based on BioNTech’s ADS closing price of $34.55 on Wednesday, January 15, 2020.
The deal has been approved by both companies’ boards of directors. It is expected to close in the second quarter of 2020.
BioNTech was founded in 2008 and focuses on personalized cancer treatments. Earlier this month the company announced publication in the journal Science data from preclinical research on its first-in-kind CAR-T cell therapeutic approach to solid tumors. The therapeutic, BNT211, is an autologous CAR-T cell therapy that targets the oncofetal antigen Claudin 6 (CLDN6). It is suggested that the company’s CARVac is a broadly applicable RNA vaccine.
CAR-T has been shown to be effective in blood cancers, but much more limited in solid tumors. BioNTech is focused on developing CAR-T therapies in multiple solid tumors in combination with an RNA vaccine.
In the published data, the therapy was studied in mice with human ovarian cancer transplants. In the research, CLDN6-CAR-T showed complete tumor regression of transplanted large human tumors within two weeks after treatment initiation. The combination with CARVac improved engraftment, proliferation and CAR-T cell expansion, all of which is promising for human studies, which the company plans to launch this year, with ovarian, testicular, uterine and lung cancer.
Of the merger with BioNTech, Hugh O’Dowd, chief executive officer of Neon, said, “We are very proud of all we have accomplished since we founded Neon and look forward to joining forces with BioNTech to continue to build a business that provides life-changing immunotherapy products to patients battling a variety of cancers.”
BioNTech Buys Kite TCR Cell Therapy Plant to Boost US Trials – Biospace 7/19/2021
Germany’s BioNTech announced it is acquiring Kite’s solid tumor neoantigen T-cell receptor (TCR) research-and-development platform and its clinical manufacturing plant in Gaithersburg, Maryland. Kite is a Gilead company.
The company is best known for its mRNA COVID-19 vaccine it developed with Pfizer. However, much of BioNTech’s pipeline is focused on using mRNA for cancer applications.
BioNTech indicates the acquisition will add production capacity in support of U.S. clinical trials. It already has a cell therapy manufacturing site in Idar-Oberstein, Germany.
The company’s pipeline includes cancer product candidates built on its CAR-T cell amplifying mRNA vaccine (CARVac) and NEOSTIM platforms in addition to the newly acquired individualized neoantigen TCR program.
“The development of individualized cancer therapies is at the core of our work at BioNTech,” said BioNTech CEO and co-founder Ugur Sahin. “It also strengthens our presence in the U.S., building on our successful integration of adoptive T-cell and neoantigen TCR therapies as part of our acquisition of Neon Therapeutics last year.”
BioNTech picked up Neon Therapeutics in an all-stock deal worth $67 million in January 2020. Neon’s focus is on neoantigen therapies for both vaccines and T-cell therapies.
Neoantigens are immune targets created by mutations inherent in tumors. Neon’s most advanced program was NEO-PTC-01, a personalized neoantigen-targeted T-cell therapy.
BioNTech’s pipeline includes BNT111 for advanced melanoma, currently in Phase II studies; BBNT112 in Phase I/II for metastatic Castration Resistant Prostate Cancer (mCRPC); and BNT113, also in Phase I studies for HPV16+ head and neck cancer, and approximately 11 others in clinical trials and more in preclinical development.
On June 18, BioNTech treated the first patient in its BNT111 Phase II trial. It is testing BNT111 in combination with Libtayo (cemiplimab) in anti-PD1-refractory/relapsed unresectable Stage III or IV melanoma. It is being run in collaboration with Regeneron Pharmaceuticals. BNT111 is an intravenous cancer vaccine that uses mRNA to encode four cancer-specific antigens. Libtayo is Regeneron and Sanofi’s anti-PD-1 checkpoint inhibitor.
“Our vision is to harness the power of the immune system against cancer and infectious diseases,” said Özelm Türeci, co-founder and chief medical officer of BioNTech at the time. “We were able to demonstrate the potential of mRNA vaccines in addressing COVID-19. We must not forget that cancer is also a global health threat, even worse than the current pandemic.”
“BNT111 has already shown a favorable safety profile and encouraging preliminary results in early clinical evaluation. With the start of patient treatment in our Phase II trial, we are encouraged to continue on our initial path to realize the potential of mRNA vaccines for cancer patients,” she continued.
Any Kite staffers working at the Gaithersburg facility will be offered employment with BioNTech. The company also indicates it plans to invest more in the location, including hiring more people. Kite has a new manufacturing site in Frederick, Maryland, to manufacture CAR T-cell therapies that are not part of this deal.
“In order to serve more patients that need cell therapy today, Kite is rapidly growing both through global expansion and seeking new indications for our existing approved CAR T-cell therapies,” said Christi Shaw, chief executive officer of Kite. “This transaction will enable us to focus our energies and investment on accelerating the reach of our current CAR-T-cell therapies and midterm pipeline.”
After signing yet another deal with COVID-19 vaccine partner Pfizer last week, BioNTech is not stopping anytime soon on the deal front. This time, the vaccine maker is partnering with Crescendo Biologics for a $40 million upfront deal to work on immunotherapies.
BioNTech will provide the upfront fee to the Cambridge, U.K.-based biotech and a potential $750 million in biobucks for immunotherapies against cancer and other undisclosed diseases. The biotech, emboldened and lined with cash from its pandemic vaccine, will choose the targets for the mRNA-based antibodies and engineered cell therapies, the company said on the first day of the annual J.P. Morgan Healthcare Conference.
The German biotech will also provide research funding and handle development in exchange for exclusive worldwide rights on any products that come out of the discovery collaboration, which has an initial timeline of three years.
The deal gives BioNTech access to Crescendo’s “Humabody” platform, which is being used to generate multi-specific therapies. The company has developed CB213, which is not part of the deal, using the platform to target T cells expressing both PD-1 and LAG-3. Humabodies retain the binding and specificity of traditional antibodies but have a smaller size with better tumor penetration and stability, BioNTech said.
The platform is aimed at retaining the binding and specificity of traditional antibodies but utilizing a smaller size with better tumor penetration and stability, BioNTech said.
“Crescendo’s platform provides excellent properties for exploiting novel targets and target combinations which we believe has great potential for the development of multi-specific mRNA and engineered cell-based therapies in a variety of disease areas,” said BioNTech CEO Ugur Sahin, M.D., in a statement.
BioNTech will join Amgen in the multi-specific game, which is a type of medicine that attacks cancer in several different ways. Several bi-specifics have been approved and are in waiting at the FDA or in clinical trials.
This is the second deal for BioNTech in five days. The company linked arms again with its Cominarty partner Pfizer last week to work on an mRNA-based shingles vaccine. Pfizer dished out $225 million upfront and will pay up to $200 million in biobucks.
BioNTech, flush with COVID-19 cash, has struck another deal. The latest agreement will see the German biotech pay 26 million euros ($29 million) for a preclinical T-cell receptor (TCR) program from its compatriot Medigene.
Through the deal, BioNTech will take control of a preclinical program targeting PRAME, an antigen that is highly expressed in several solid tumors but largely limited to the testis in healthy tissues. The expression profile of PRAME led Medigene to genetically modify T cells to express both a TCR against the antigen and a PD1-41BB switch receptor designed to prevent inhibition in the tumor microenvironment.
BioNTech is acquiring the program as part of a deal that also gives it exclusive options on other TCRs. The partners will collaborate on the development of TCRs, with Medigene applying its discovery platform to multiple solid tumor targets selected by BioNTech under the terms of a three-year partnership.
In return, BioNTech is paying 26 million euros upfront, plus research funding for the collaboration, and committing to milestones that could top 100 million euros per program. The outlay also gives BioNTech licenses to Medigene’s PD1-41BB switch receptor and precision pairing library for use across its cell therapy programs.
Those cell therapies have played second fiddle as BioNTech’s mRNA capabilities have put it on the map, but they are a key area of focus for the biotech. Last year, BioNTech struck a deal to buy a solid tumor neoantigen TCR R&D platform and clinical manufacturing facility from Gilead’s Kite Pharma to add to its existing capabilities in Germany. The Medigene deal fits into the same strategy.
BioNTech has also used its cash reserves, which came in at 2.4 billion euros and climbing at the end of the third quarter, to enter into a collaboration with Crescendo Biologics to develop engineered cell therapies as well as mRNA-based antibodies. The string of cell therapy deals was only broken up by the takeover of antibacterial biotech PhagoMed Biopharma for 50 million euros upfront in October.
The addition of Medigene to BioNTech’s list of cell therapy deals sent investors into overdrive. Shares in Medigene soared more than 90% to around 4 euros in early trading. While Medigene’s stock has hovered around the 4-euro mark for much of the past year, it fell to below 2 euros earlier this month. The BioNTech deal marks a rare moment in the spotlight for a biotech now perhaps better known for spawning Adaptimmune and Immunocore than it is for its internal achievements.
BioNTech has struck a deal to explore oral delivery of mRNA vaccines. Working with Matinas BioPharma, the German mRNA specialist will study the potential for a lipid nanocrystal (LNC) platform to enable new vaccine formulations.
The pandemic catapulted BioNTech into the biotech big leagues as the vaccine it discovered and then developed with Pfizer became a cornerstone of vaccination COVID-19 campaigns around the world. From that position of strength, BioNTech has identified Matinas as a company that can help it build a business beyond the Comirnaty windfall.
BioNTech is paying $2.75 million for exclusive access to Matinas’ LNC platform in the delivery of mRNA vaccines, and will fund some of its new partner’s research expenses related to the collaboration. Talks about a potential licensing agreement are underway.
The deal gives BioNTech a chance to take a closer look at the value the platform could bring to its mRNA vaccines. Matinas, which also has a LNC partnership with Genentech, licensed the platform from Rutgers University to enable the targeted intracellular delivery of a range of molecules, including mRNA.
In the context of mRNA vaccines, the stability of LNC structures has potentially significant implications. According to Matinas, the highly stable structure “allows for the avoidance of extreme cold chain storage temperatures required for maintaining the integrity of [lipid nanoparticles],” such as those used in the administration of Comirnaty. The Pfizer-BioNTech COVID-19 vaccine is stored at -90°C to -60°C.
If the LNC platform can enable mRNA vaccines to be stored at higher temperatures, it would be easier to get the products to people in the developing world, although the pandemic has already increased access to ultracold chain freezers.
The stability of the LNC structure also opens the door to oral administration. Matinas is already using the platform to enable oral administration of its own molecules and BioNTech wants to assess whether it can open up a new route of delivery of mRNA vaccines.
It may have become a household name during the pandemic, but the drumbeat of announcements from BioNTech this year are a reminder that the German company has far more varied ambitions. With infectious disease and cancer vaccines in various stages of development, the biotech has now licensed a variety of small-molecule programs from Poland’s Ryvu Therapeutics.
Under the multi-target agreement, BioNtech will pay Ryvu 20 million euros ($20.7 million) upfront along with an equity investment of the same amount. Split into two parts, one side of the deal will see BioNTech secure an exclusive global license to develop and commercialize Ryvu’s STING agonist portfolio as standalone small molecules, including as monotherapies and in combination with other therapies.
The stimulator of interferon genes (STING) pathway is an immunity pathway that plays a key role both in responding to infections and autoimmune diseases as well as providing antitumor immunity. Preclinical studies have shown that Ryvu’s STING agonists were able to activate proinflammatory cytokine production and long-lasting immune responses, BioNTech said in a release.
The second part of the agreement will see the two companies jointly undertake drug discovery and research projects to develop multiple small-molecule programs for targets selected by BioNTech. The primary focus of these targets will be immune modulation for oncology, but the biotech has the option to push into other disease areas. Once candidates have been singled out for development, BioNTech will have the option to license them.
BioNTech will fund all discovery, research and development activities, with Ryvu also in line for undisclosed milestone payments and low single-digit royalties.
“Small molecules targeting novel immune signaling pathways have a great potential to increase the efficacy of cancer immunotherapies,” said BioNTech CEO Ugur Sahin, M.D., in a Nov. 30 release. “The collaboration with Ryvu provides us with the opportunity to complement our immunotherapy pipeline with a portfolio of potent immunomodulatory molecules.”
BioNTech’s oncology pipeline is already well underway. The company has 19 candidates across a total of 24 clinical trials, including CAR-T cell therapy candidate BNT211. The furthest developed are phase 2 trials of melanoma vaccine BNT111 and BNT113 for HPV16-positive cancers—which both use the biotech’s FixVac platform—plus a pancreatic cancer hopeful called autogene cevumeran and the bispecific antibody immune checkpoint modulator BNT311.
A number of drug developers have been stung in their attempts to chase the STING pathway. In 2019, Novartis ended work on an intratumoral STING pathway activator candidate developed by Aduro Biotech, while Nimbus Therapeutics also pivoted away from the pathway the following year.
BioNTech isn’t the only one who still sees potential, however. AstraZenecasnagged the global rights to research, develop and bring to market STING inhibitor compounds for $12 million upfront from F-star Therapeutics last year.
While BioNTech is still keeping one foot in the COVID-19 arena that made its name, the German company has made a concerted push toward treating cancer in recent months.
That effort took another leap forward Monday as the biotech announced a licensing deal with OncoC4, taking on the latter’s mid-stage CTLA-4-targeting monoclonal antibody, ONC-392, for $200 million in upfront cash. The exclusive licensing and collaboration agreement also includes an undisclosed amount of commercial and clinical milestones.
The deal is evidence of BioNTech’s fervor to keep its foot on the clinical gas pedal, particularly in cancer. ONC-392 is currently being assessed in a phase 1/2 trial in patients with solid tumors as both a monotherapy and combination treatment alongside Merck & Co.’s blockbuster Keytruda. Another phase 2 trial is looking at the same combo treatment in patients with platinum-resistant ovarian cancer.
The data accrued from the ongoing phase 1/2 trial supports the launch of a phase 3 trial testing ONC-392 as a monotherapy in patients with PD-L1-resistant non-small cell lung cancer, BioNTech said in the release.
The company believes the newly-acquired antibody has a “differentiated safety profile,” CEO Ugur Sahin, M.D., said. “Despite being a prime target for more than a decade, we believe that targeting CTLA-4 has not reached its full potential in cancer immunotherapy.”
The knock on the target in the past has been a significant level of immune-related adverse events. One systemic review from 2015 concluded that the tradeoff of anti-CTLA4 antibodies was “atypical immune toxicity.”
ONC-392 slots into an ever-growing cancer pipeline for BioNTech, which includes nearly two dozen clinical-stage assets, five of which are antibodies. Four of the antibodies are part of a collaboration with Genmab, including phase 2 BNT311 currently targeting patients with metastatic non-small cell lung cancer.
Much of the star power still rests with BioNTech’s cancer vaccine research, particularly after the U.K. government partnered with the company to deliver 10,000 personalized therapies by 2030. As part of the agreement, BioNTech will build a new R&D hub in the country, expanding its European footprint.
BioNTech moves into ADCs with Duality deal – Biopharmadive 4/3/2023
Dive Insight:
Armed with cash earned from sales of the COVID-19 vaccine it developed with Pfizer, BioNTech has been investing in cancer drug research and development — its primary focus prior to the pandemic.
While BioNTech specializes in messenger RNA, it also has built a pipeline of antibody drugs, too. Most recently, it spent $200 million for rights to a cancer immunotherapy from the biotechnology company OncoC4.
The deal with Duality adds ADCs to BioNTech’s pipeline, taking the company into a fast-growing field. ADCs pair a cell-killing toxin with a targeting compound designed to home in on tumors while sparing healthy tissue. The complex drugs have a long history, but pharmaceutical companies’ interest in them has grown over the past several years, buoyed by clinical trial successes.
The Food and Drug Administration has approved eight ADCs since 2019, including AstraZeneca and Daiichi Sankyo’s breast cancer drug Enhertu. Dramatic study results for that treatment led the agency to expand its approval last summer to include people whose tumors have low levels of a protein called HER2.
Still, while ADCs have shown promise, they can cause severe side effects as well.
Duality’s lead candidate, dubbed DB-1303, also targets the HER2 protein, which is overexpressed in breast cancer and a number of other tumors. The drug is currently in a Phase 2 clinical trial for advanced solid tumors that are positive for HER2.
The other candidate BioNTech gains, DB13-11, is in preclinical development for “multiple cancer types.”
BioNTech isn’t the first Western drugmaker to turn to China-based biotechs for ADC technology. In 2021, Seagen — a pioneer in the field — struck a similarly sized deal with the Yantai, China-based biotech RemeGen to gain access to a HER2-targeting ADC.
BioNTech has swiftly rejected an oral mRNA delivery technology. Barely one year after signing up to work with Matinas BioPharma, BioNTech found an initial mouse study failed to show preclinical activity—leading it to join Gilead Sciences on the list of companies to dump the biotech in recent months.
In April 2022, Matinas revealed BioNTech had paid $2.75 million for exclusive use of a lipid nanocrystal (LNC) platform in the delivery of mRNA vaccines. BioNTech agreed to fund Matinas’ research related to the collaboration, which covered formulation, optimization and in vitro testing, and entered into talks about a potential licensing agreement.
The collaboration made it as far as a study in mice. Thursday, Matinas revealed that the study of the oral mRNA candidate failed to show activity. With the single study now over, BioNTech and Matinas have ended their collaboration.
Matinas’ summary of the collaboration offers insights into the challenges of delivering mRNA orally. The April 2022 statement about the deal said the collaboration would “evaluate the combination of mRNA formats and Matinas’ proprietary LNC platform technology.” However, the mouse study tested a non-LNC formulation of a BioNTech-supplied reporter mRNA.
As Matinas explained, it developed a “nano-formulation, distinct from traditional LNCs” to cope with the “physical complexity and biological fragility of mRNA.” The formulation worked in vitro, Matinas said, “and because of the timelines required under the BioNTech collaboration was brought forward for oral in vivo evaluation.”
While BioNTech is walking away, Matinas made the case that its approach may work, stating that internal in vivo studies of similar non-LNC mRNA formulations “showed activity when administered systemically.” The formulations are stable for at least 17 weeks at 4 degrees Celsius, suggesting they may be easier to ship and store than existing mRNA vaccines based on lipid nanoparticles.
The conclusion of the BioNTech deal comes five months after Gilead pulled out of a deal with Matinas to develop an oral COVID-19 antiviral to focus on its internal candidate. Roche’s Genentech unit recently extended its alliance with Matinas for another year, although, with the biotech completing its obligations related to a third and final molecule in the first quarter, the longer-term prospects remain uncertain.
BioNTech acquires tech company InstaDeep for $549m – Pharmaceutical Technology 8/1/2023
BioNTech has acquired all the remaining shares of technology company InstaDeep in a deal valued at €500m ($549m) in cash. The total deal value excludes the shares BioNTech already owns.
In January 2023, the parties signed an agreement for the acquisition.
UK-based InstaDeep is a technology provider in the fields of artificial intelligence (AI) and machine learning (ML). The takeover follows a collaboration between the two parties since 2019 and an equity investment by BioNTech in InstaDeep through a Series B funding round held in January 2022.
BioNTech expects that the deal will help it establish robust expertise in AI-powered drug discovery and the development of advanced vaccines and immunotherapies for conditions with high unmet medical needs.
BioNTech has already been tinkering with its manufacturing processes in the run up to CAR-T BNT211 entering pivotal trials. Now, the German biotech has brought British company Autolus Therapeutics on board in an intriguing $250 million upfront collaboration.
The deal means BioNTech can use Autolus’ manufacturing and clinical site network across the U.K. to help efficiently develop BNT211 for pivotal trials in CLDN6+ tumors, the companies said in a Feb. 8 release.
In addition, BioNTech gains an exclusive license to use certain target binders identified by U.K.-based Autolus as well as the option to license additional binders or cell programming technologies to support the German biotech’s own in vivo cell therapy and antibody-drug conjugate (ADC) candidates. Autolus will be eligible to receive milestone payments from any resulting drugs.
In return, BioNTech will support the launch and expansion of Autolus’ lead autologous CD19 CAR-T, dubbed obe-cel, which is awaiting an FDA decision for patients with B-cell acute lymphoblastic leukemia.
BioNTech will also gain co-commercialization options for Autolus’ AUTO1/22 program, which has undergone a phase 1 study for relapse in patients who have received a CAR T-cell therapy for B-cell acute lymphoblastic leukemia, as well as AUTO6NG, which was expected to enter the clinic late last year for children with neuroblastoma.
BioNTech described the agreement as a “strategic collaboration aimed at advancing both companies’ autologous CAR-T programs towards commercialization, pending regulatory authorizations.”
While it appears that both biotechs will benefit from the collaboration, Autolus’ bank balance definitely wins out. BioNTech is not only handing over $50 million cash but will also buy $200 million of Autolus’ American depositary shares (ADSs) in a private placement. In return, BioNTech gets the right to appoint a director to Autolus’ board and is eligible for up to mid-single digit royalties on obe-cel sales.
To coincide with the BioNTech announcement this morning, Autolus said it was making a public offering of a further 58.3 million ADSs, which are expected to bring in gross proceeds of $350 million.
Autolus CEO Christian Itin said the deal with BioNTech was “a remarkable opportunity to leverage our core capabilities, accelerate pipeline programs, realize cost-efficiencies and expand opportunities beyond autologous cell therapies.”
Back in August, BioNTech executives attributed part of the success of the CLDN6 CAR-T program BNT211 in a solid tumor clinical trial to the company’s new manufacturing process. In today’s release, the biotech said it plans to have “10 or more ongoing potentially registrational clinical trials in the pipeline by the end of 2024,” which include BNT211 in relapsed or refractory germ cell tumors.
“The collaboration with Autolus enables us to expand our BNT211 program into trials for multiple cancer indications in a cost-efficient way,” BioNTech’s CEO Ugur Sahin, M.D., said in today’s release. “Autolus’ state-of-the-art manufacturing facilities’ set-up for clinical and commercial supply will enhance our own capacities in addition to our existing U.S. supply network and the ongoing expansion of our site in Gaithersburg, Maryland.”