(Picture: Alex Zhavoronkov, Ph.D., founder and CEO of Insilico Medicine)
Boston Dr. Lim is here. Alex Zhavoronkov was a computer scientist but became interested in aging and computer science. In order to study aging, he quit his job and went to Johns Hopkins University to get master degree of biotechnology and finally got PhD in biophysics in Moskow University. In 2011, Alex and Charles R. Cantor published a PLOS One paper demonstrating how artificial intelligence could be used for aging research. This paper became the foundation to start “Insilico Medicine” in 2014. Since the founding, Insilico Medicine raised more than $400 million through multiple rounds and delivered novel small molecules against novel therapeutic targets to human clinical trials. The most advanced product is INS017_055 targeting TNIK to treat fibrosis. So far 9 preclinical novel molecules can be nominated every year and goal is to deliver 15 molecules a year. I expect both INS018_055 and other novel generative AI-generative drugs could finish the FDA approval line in the end. Moreover, Alex would like to tackle clinical testing with generative AI. I wonder how Insilico Medicine team could crack the code. Insilico Medicine is global company.
GlaxoSmithKline taps Baltimore’s Insilico for AI-based drug discovery – Fierce Biotech 9/16/2017
GlaxoSmithKline and Insilico Medicine are partnering to explore how the latter’s artificial intelligence technology can aid in the drug discovery process.
The duo kept mum on the details but said the agreement comes after Baltimore-based Insilico completed a series of pilot challenges. First, Glaxo will “evaluate Insilico’s technology in the identification of novel biological targets and pathways of interest,” according to a statement.
Based at Johns Hopkins University’s Emerging Technology Centers, Insilico uses genomics, big data analysis and deep learning for in silico drug discovery, or drug discovery through computer modeling. The company has its own drug discovery programs in cancer, amyotrophic lateral sclerosis and diabetes, as well as in age-related diseases such as sarcopenia, Parkinson’s disease and Alzheimer’s disease.
“We are delighted to be working with the Insilico team, as they have exhibited curiosity, agility and AI expertise that we value,” said John Baldoni, senior vice president of platform technology and science at GSK, in the statement. “GSK recently established a drug discovery unit to explore how this rapidly developing field might drive drug discovery at a higher velocity, with greater precision and at a reduced cost. The collaboration with Insilico represents one of several approaches we are exploring to take advantage of emerging technology that might make us more effective and efficient, always keeping in mind the patients who need new medicines.”
RELATED: BenevolentAI looks to artificial intelligence for speedy development of in-licensed drugs
The collaboration is GSK’s second drug discovery deal of the summer based around AI. In July, the Big Pharma inked a deal with Exscientia—which automates drug discovery with its AI-enabled platform—that could see the Dundee, U.K.-based company rake in up to £33 million ($42.7 million) in research payments. The Exscientia partnership aims to identify small molecules to treat as many as 10 targets chosen by GSK.
“In our opinion, GSK is one of the most innovative science-led healthcare companies, which realized the potential of artificial intelligence early and has demonstrated its ability to partner with innovative startups in the field. We are delighted to collaborate with arguably, some of the world’s best scientists on chronic diseases with unmet need,” Alex Zhavoronkov, Ph.D., founder and CEO of Insilico Medicine, said in the statement.
WuXi AppTec to test AI-generated compounds from Insilico Medicine – Fierce Biotech 6/11/2018
WuXi AppTec is partnering up with Insilico Medicine through a venture capital investment to boost its artificial intelligence-driven drug and biomarker discovery platforms.
While a smaller, previous research collaboration with WuXi AppTec has been ongoing, the new agreement represents a larger push to validate and optimize Insilico’s pipeline, with plans to formally integrate the Baltimore-based company’s technology with WuXi AppTec’s laboratory infrastructure.
Insilico’s generative adversarial networks employ two, competing neural networks to produce and evaluate candidates and drug targets through a machine learning-based discovery pipeline. The total investment amount was undisclosed.
The transaction was a joint effort between WuXi AppTec’s Research Services Division and Corporate Venture Fund, which led the VC funding round. BOLD Capital Partners and Pavilion Capital, a subsidiary of Temasek Holdings, also participated in the financing—as well as Juvenescence, a longevity therapeutics company that holds a discovery partnership with Insilico.
“WuXi AppTec and Insilico Medicine share a mutual vision that artificial intelligence and machine learning will optimize the drug discovery process by increasing the probability of success at the pre-clinical level,” WuXi AppTec Chairman Ge Li said in a statement.
“Insilico Medicine’s domain expertise in next-generation artificial intelligence, coupled with WuXi AppTec’s capability platform, can potentially improve the efficiency of drug discovery and increase the productivity to serve our partners,” Li added.
Under the agreement, the companies will perform a series of experiments, where novel molecules discovered at Insilico will be tested at WuXi AppTec. A series of milestones apply to preclinical candidates in challenging biological targets, including orphan diseases with no known crystal structure and no known ligands.
RELATED: GlaxoSmithKline taps Baltimore’s Insilico for AI-based drug discovery
“Our collaboration with WuXi AppTec enables us to focus on artificial intelligence without the need to invest in the expensive laboratory infrastructure,” said Alex Zhavoronkov, Insilico’s founder and CEO.
“More than 90% of the molecules discovered the traditional techniques and tested in mice fail in human clinical trials. Our goal at Insilico Medicine is to develop advanced end-to-end AI solutions to discover the optimal preclinical candidates,” Zhavoronkov said.
Insilico plans to use the funding to build upon its work in novel molecule discovery, as well as expand its biomarker initiatives.
RELATED: WuXi AppTec boosts U.S. footprint with expanded New Jersey lab site
“We have been looking to collaborate with artificial intelligence-enabled technologies to enhance the drug discovery and development process,” said Edward Hu, WuXi AppTec’s chief financial officer and chief investment officer.
“What impressed us the most about Insilico Medicine was the breadth of its technological capabilities and applications, ranging from target identification and small molecule generation using next-generation artificial intelligence to expertise in blockchain and aging research.”
AI-based drug designer Insilico Medicine has raised $37 million to help commercialize its technology, on the heels of a landmark paper for the company that showed its computer networks were able to generate, synthesize and preclinically validate a series of promising compounds from scratch in less than 50 days.
Insilico also plans to build out its senior management team. Specifically, it’s angling for industry veterans that could help secure discovery partnerships with biopharmaceutical companies across different therapeutic programs, including in cancer, immunology, fibrosis, nonalcoholic steatohepatitis and central nervous system conditions.
The series B round was led by China-based Qiming Venture Partners, with participation from Eight Roads, F-Prime Capital, Lilly Asia Ventures, Sinovation Ventures, Baidu Ventures, Pavilion Capital, BOLD Capital Partners and others, including longevity-focused company Juvenescence.
“The company is an industry leader in the AI-powered drug discovery vertical,” Qiming Managing Partner Nisa Leung said in a statement. “We look forward to seeing it shortening the time for drug discovery and creating synergies with our portfolio companies.”
Another of the company’s investors—Deep Knowledge Ventures, which has backed Insilico since its 2014 seed round—described its work as one of the fund’s most promising, and a central player among its companies focused on AI-based drug discovery and longevity.
Additionally, the firm is currently launching two new subsidiary funds in those two areas—dubbed AI-Pharma and Longevity.Capital—according to Deep Knowledge Managing Partner Margaretta Colangelo, who described (PDF) them as “hybrid investment funds” combining the features of venture and hedge funds to help derisk projects.
RELATED: WuXi AppTec to test AI-generated compounds from Insilico Medicine
Earlier this month, Insilico had its work published in Nature Biotechnology, demonstrating how machine learning networks could potentially shave years off of traditional hit-to-lead timelines for drug development.
In 21 days, the company was able to conceptualize 30,000 novel small molecules against a target linked to fibrosis. Over 25 more, Insilico and its partners synthesized the six most promising and tested them in both in vitro and live mouse models.
Within 25 more days, they had screened out and synthesized the six most promising compounds, tested them in vitro for selectivity and metabolic stability and had the lead candidate go on to show favorable activity in live mouse models.
The advent of artificial intelligence holds the promise to reset the industry’s expectations of how long it should take to develop a new drug from scratch. And with serious new investments, Insilico Medicine is looking to make it an everyday reality.
The company, with operations straddling the U.S. and Hong Kong, raised $255 million in series C funding led by Warburg Pincus—a big step up from its last round, which topped just $37 million, in 2019—serving as a reflection of what it has achieved in the intervening years.
That year, Insilico showed its AI systems could generate tens of thousands of novel molecules for a particular target, whittle them down to the most promising candidates and demonstrate promising drug activity in mouse studies of the disease, all in less than 50 days.
And this past February, the former Fierce 15 winner reported the completion of in vitro and in vivo preclinical studies—in a span of fewer than 18 months and with a total price tag of about $2 million—for an oral compound with a brand new mechanism of action that aims to improve lung function in patients with pulmonary fibrosis.
Insilico has also commercially launched its PandaOmics AI engine for target discovery, its Chemistry42 system for molecule design and its InClinico service for predicting clinical trial outcomes—while maintaining collaborations with Pfizer, Astellas, Johnson & Johnson, Taisho and other international drugmakers. And outside of human medicine, it’s partnered with agriculture company Syngenta to develop new molecules for more sustainable farming techniques.
Now, with its hefty proceeds, the company plans to move its therapeutic programs forward into human clinical trials as well as launch research programs for additional novel targets.
RELATED: Insilico Medicine taps GSK alum for a 6-month AI sprint aimed at brain cancer
The series C round included new backing from CPE, OrbiMed, Mirae Asset Capital, B Capital Group, Deerfield Management, Maison Capital, Lake Bleu Capital, President International Development Corporation, Sequoia Capital China and Sage Partners as well as funds from its previous investors Qiming Venture Partners, Pavilion Capital, Eight Roads Ventures, Lilly Asia Ventures, Sinovation Ventures, BOLD Capital Partners, Formic Ventures and Baidu Ventures.
The funding total follows other large venture capital hauls in the AI drug prospecting space, including insitro’s massive $400 million round this past March and Atomwise’s $123 million draw last year.
More recently, SoftBank led a funding round for Exscientia that could net it up to $525 million in financing and equity commitments. Meanwhile, Recursion Pharmaceuticals ramped up its IPO to just over $500 million by the time it closed.
Insilico Medicine, a clinical-stage end-to-end artificial intelligence (AI)-driven drug discovery company, announced today that it has completed a $60 million Series D financing from a syndicate of global investors with expertise in investing in the biopharmaceutical and life sciences sectors.
New investors, including a large, diversified asset management firm on the US West Coast and BHR Partners joined the round, along with current investors, including lead investor of Series C financing round Warburg Pincus, B Capital Group, Qiming Venture Partners, BOLD Capital Partners and Pavilion Capital. Insilico’s founder and CEO, Alex Zhavoronkov, PhD, also invested in the Series D round.
Capital raised in the round will further bolster Insilico’s financial position and fuel the growth of its advancing pipeline, including its lead program which is currently in a Phase I study, and continued development of its Pharma.AI platform. The proceeds will also fund ongoing global expansion and planned strategic initiatives, including a fully automated, AI-driven robotic drug discovery laboratory, and fully robotic biological data factory to complement Insilico’s vast curated data assets.
“Despite unprecedented market conditions in the biotechnology sector, we raised this Series D round from several of the most reputable US-based and global investors,” said Alex Zhavoronkov, PhD, Founder and CEO of Insilico Medicine. “It is a testament to the strength of our end-to-end AI platform, which has been validated by many partners, and produced our first novel antifibrotic program discovered using AI and aging research, and designed using our generative AI chemistry engine. This unique program completed a first-in-human Phase 0 study in healthy volunteers and has entered Phase I clinical trials. We have also nominated seven preclinical candidates across a number of other disease indications since 2021. I am very excited about this progress and have decided to personally invest in this round.”
“The application of artificial intelligence and machine learning for drug discovery has incredible potential to transform the way new therapies are developed,” said Min Fang, Managing Director, Head of China Healthcare at Warburg Pincus. “For Insilico, 2022 is a year of incredible growth and progress. They have demonstrated the value of combining deep scientific expertise with cutting-edge technology capabilities to significantly accelerate drug discovery. We’re delighted to continue to partner with the Insilico team and support a company that is at the forefront of this innovation.”
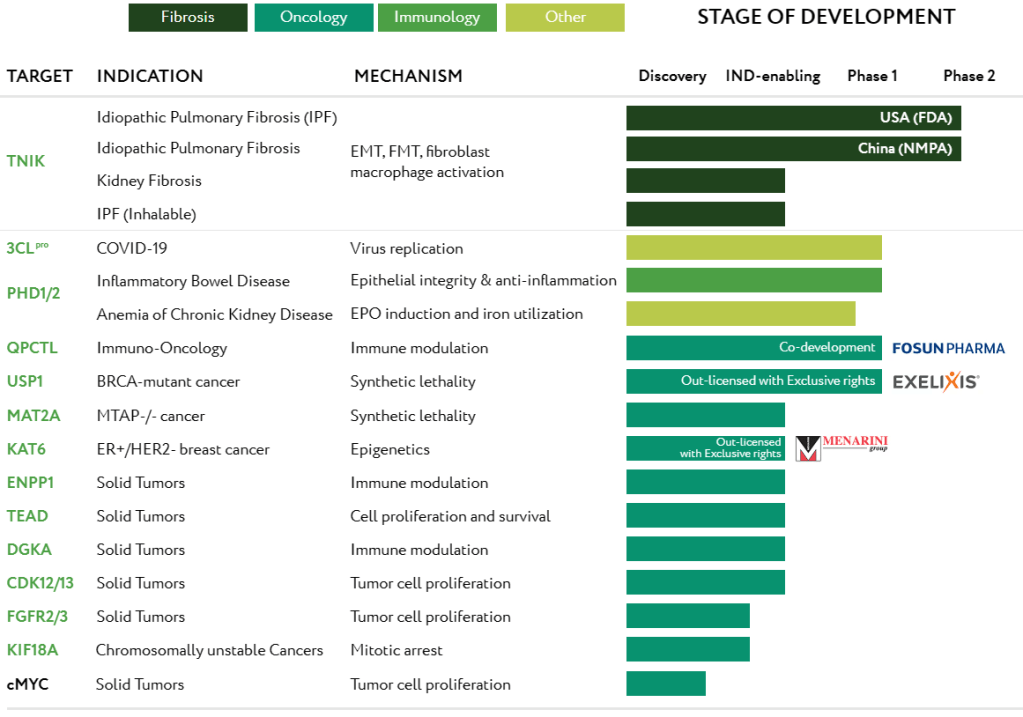
Since the previous round of financing, Insilico has developed a growing portfolio in frontier areas empowered by its proprietary AI platform. Seven programs in its internal pipeline have progressed to the IND-enabling stage, including a novel 3CL protease inhibitor for COVID-19 treatment, and two synthetic lethality programs targeting MAT2A and USP1 for oncology. It also successfully completed a Phase 0 microdose study and entered a Phase I clinical trial with its first internally developed program targeting fibrosis.
“With the power of cutting-edge AI platforms developed fully in house and validated by many global pharmaceutical companies and our innovative, highly parallel, fully distributed drug discovery model, we nominated seven preclinical candidates since I joined the company in 2021,” said Feng Ren, PhD, Insilico’s Chief Scientific Officer and Head of Global Research and Development. “I have 15 years of experience working in large pharmaceutical companies and CROs and I am impressed by the speed and quality of our discovery programs. We plan to continue expanding the breadth of the pipeline and enhance our AI and robotics capabilities globally. Our rapidly growing team is composed of talented and experienced scientists in drug discovery with diverse backgrounds and relentless passion for novelty and innovation, and fully committed to developing novel drugs with a sense of urgency for the waiting patients.”
Insilico has also expanded its collaborations with the pharmaceutical industry through co-development and software licensing deals with a number of major pharmaceutical companies. Since the launch of its PandaOmics™ and Chemistry42™ in late 2020, nine out of the top 20 pharmaceutical companies have licensed Insilico’s AI platforms. In 2022, Insilico signed a multi-asset partnership deal with Fosun Pharma in January, and a drug discovery co-development deal with EQRx in March. Notably, Insilico achieved its first major milestone and nominated a preclinical candidate for the QPCTL program for cancer immunotherapy in less than 40 days into the strategic collaboration with Fosun Pharma.
About Insilico Medicine
Insilico Medicine, a clinical stage end-to-end artificial intelligence (AI)-driven drug discovery company, is connecting biology, chemistry, and clinical trials analysis using next-generation AI systems. The company has developed AI platforms that utilize deep generative models, reinforcement learning, transformers, and other modern machine learning techniques to discover novel targets and to design novel molecular structures with desired properties. Insilico Medicine is delivering breakthrough solutions to discover and develop innovative drugs for cancer, fibrosis, immunity, central nervous system diseases and aging-related diseases.
Alex Zhavoronkov Aims to Take Over the Drug Development World with AI – GEN Edge 6/20/2023
In Suzhou, a city 30 minutes by train from Shanghai, there’s a nondescript building that looks like it’s covered in Death Star surface tiles. In the middle of the building’s facade is a logo of a square computer chip with an Erlenmeyer flask in the middle. The logo seems to glow the color of green battery-charging lights.
A virtual walkthrough starts at the lab entrance. Like a scene from Mission Impossible, the doors are primed to open with facial recognition followed by touch activation on the airlock-like, floor-to-ceiling door. If the video weren’t on mute, I wouldn’t be surprised to hear hydraulic and whooshing sounds.
“I want to make you feel like you’re in Star Wars,” Alex Zhavoronkov, co-founder and co-CEO of Insilico Medicine, told me while showing real-life footage of his fully automated laboratory from his laptop.
As the camera guided me through the futuristic doors of his real-life sci-fi lab—Zhavoronkov has dubbed it Life Star—it pans into a glass room the size of a tennis court, with mechanical arms swinging and autonomous mobile robots shuttling around. “Here the magic happens,” smiled Zhavoronkov.

The robot works with various types of samples—cells, tissues, or organoids—prepping them for various kinds of imaging and next-generation sequencing techniques, including those for profiling DNA, RNA, and methylation. After the robotic ballet finishes generating data, the data goes back to Insilico’s trademark artificial intelligence (AI) for target selection. If anything interesting shows up, scientists will begin a validation process.
Life Star is functional around the clock. And every one of Insilico Medicine’s programs is now supported in some way by the autonomous, AI-powered robotics laboratory. Construction for his robotic laboratory in China began during the COVID-19 pandemic. “The government allowed us to bubble there, so people lived there!” Zhavoronkhov said, showing me a picture of himself lying on what looked like a gurney in a room covered with plastic. “I slept there for four months.”
The scientist
Zhavoronkov, whose real name is Aleksandrs Zavoronkovs (according to his Twitter), is unabashedly ambitious and brilliant; he’s a borderline mad scientist. He doesn’t fit the sci-fi motif of an evil genius that wants to take over the world (although Insilico Medicine has sites in Suzhou, Shanghai, Hong Kong, Taipei, Abu Dhabi, New York, San Francisco, and Montreal).
Under his leadership, Insilico has raised a total of $401.3M in funding over 10 rounds. Their latest funding was raised on Aug 10, 2022, from a second Series D round led by Prosperity7 Ventures and Aramco Ventures, bringing the total Series D financing to $95 million. New investors, including a large, diversified asset management firm on the U.S. West Coast and BHR Partners joined the round, along with current investors, including lead investor of Series C financing round Warburg Pincus, B Capital Group, Qiming Venture Partners, BOLD Capital Partners, and Pavilion Capital. Zhavoronkov’s fundraising has allowed him to take Insilico global, having opened several R&D centers around the world, and partnered with multiple pharmaceutical, biotechnology, and academic institutions.
Zhavoronkov holds two bachelor’s degrees from Queen’s University, a Master’s degree in biotechnology from John Hopkins University, and a PhD in physics and mathematics from Moscow State University. Since 2012, he has published over 150 peer-reviewed research papers, and two books including “The Ageless Generation: How Biomedical Advances Will Transform the Global Economy.” Zhavoronkov is an adjunct professor of artificial intelligence at the Buck Institute for Research on Aging. In addition to serving on the advisory or editorial boards of various journals and co-chairing the annual Aging Research and Drug Discovery conference, Zhavoronkov writes articles for Forbes in his free time.
The real McCoy
In the past year, Insilico nominated a handful of preclinical candidates, and generated positive topline Phase I data in human clinical trials with an AI-discovered novel target and AI-designed novel molecule for idiopathic pulmonary fibrosis that received Orphan Drug Designation from the FDA and is nearing Phase II clinical trials. Insilico also recently announced that its generative AI-designed drug for COVID-19 and related variants have been approved for clinical trials, as has the company’s USP1 inhibitor for the treatment of patients with solid tumors.
This past year, Insilico Medicine has used its end-to-end AI-backed drug discovery and development pipeline to nominate nine preclinical candidates in small molecules, which Zhavoronkov thinks can be pushed up to fifteen a year.

Of these, Insilico Medicine was able to advance through preclinical trials and into Phase I in about one year. To put that in perspective, recent research from McKinsey shows that, over the past decade, the average time to take a new medication from candidate nomination for preclinical testing to first-in-human trials has been about 26 months. Additionally, for a pharma company seeking to move three to five investigational new drugs into first-in-human studies every year, an acceleration down to a year, applied across the portfolio, could translate into a risk-adjusted net present value exceeding $400 million.
Zhavoronkov, who is very matter-of-fact, has a bone to pick with most people who say they run AI companies for drug discovery and development. “The company should be using deep learning technology to some extent,” said Zhavoronkov. “For me to consider somebody as an AI company, I would really need to see a substantial AI component, like deep learning and generative AI, and the company should at least to some extent contribute to the development of the software field, not just use somebody else’s tools.”
And then there are those who, Zhavoronkov said, claim to be AI companies, but they are just users and those who are doing high-level googling or Excel with advanced algorithms. “I have never seen an incubator company produce a genuine AI product from an AI system,” said Zhavoronkov.
“I’m not going to name them, but usually the founder of these companies was actually not exactly in the field right away.” Successful VC companies like Flagship Pioneering and Foresight Capital were created to access financial markets, he said. “Some of [these companies] missed the opportunity to list because 2021 was the year of abundance. Everybody got enormous amounts of money and huge valuations. Some companies jumped onto the market prematurely, some timed it extremely well, and some did not list. We’re in 2023! Show me a single asset! Lots of hype, but no results. So, the industry has consolidated.”
Zhavoronkov thinks that the resulting environment has been tough on young entrepreneurs who are getting into the field and have good ideas and algorithms, but cannot fundraise because investors have seen their money go up in flames.
“Drug discovery programs are usually like $50 million to scratch the surface; I was shocked,” said Zhavoronkov. “So you raise $400 million and burn half of that in the first year on multiple programs. If you create something great, you can actually generate potential revenue. If you don’t, you’re screwed.”
According to Zhavoronkov, there are only a handful of AI-powered drug discovery and development companies in addition to Insilico Medicine. He cited Recursion and Benevolent AI, although the latter recently announced significant layoffs. If you ask ChatGPT (with data up to 2021) to name two AI-powered drug discovery companies, Insilico Medicine routinely shows up at the top. “I think that we show up there because we publish in this field one or two research papers a month on AI,” he said.
Two are better than one
The reason Insilico Medicine is successful, according to Zhavoronkov, boils down to two reasons. First, they developed a complete end-to-end AI platform in the first few years called PHARMA.AI. This drug discovery engine utilizes millions of data samples and multiple data types to discover signatures of diseases and identify the most promising targets for billions of molecules that already exist or can be generated de novo with preferred sets of parameters. This suite was created to accelerate three areas of drug discovery and development: disease target identification (PandaOmics), generation of novel molecules (Chemistry42), and predicting clinical trial outcomes (inClinico).
Despite the popularity of ChatGPT, Zhavoronkov, not surprisingly, has created something better for anyone asking research questions by text: ChatPandaGPT. This software has integrated advanced AI chat functionality based on recent advances in large language models into its PandaOmics platform. ChatPandaGPT enables researchers to have natural language conversations with the platform and efficiently navigate and analyze large datasets, facilitating the discovery of potential therapeutic targets and biomarkers in a more efficient manner. Insilico Medicine is the first biotech company to implement chat functionality using large language models into its AI drug discovery platform.
Zhavoronkov next shows another video—a sped-up example of using Insilico Medicine’s end-to-end platform. After picking an indication, which Zhavoronkov said is purely based on commercial purposes and can be done with the help of AI, he runs through a use-case of target identification and drug discovery. All it requires is clicking and patience, as it takes hours, even days, to run some of the computations.
Second, Zhavoronkov has a co-CEO, Feng Ren, PhD, whom he said is a real drug hunter. Zhavoronkov met Ren in 2020, while he was at Medicilon, a contract research organization (CRO) providing drug discovery services to biopharmaceutical companies globally. Ren served as Medicilon’s senior vice president and head of the drug R&D service business, with more than 600 chemists and biologists. “He knew how to discover drugs, but he didn’t want to provide services to the others,” said Zhavoronkov. “He wanted to really discover, and he left a lot of money on the table.”
Ren is now using 80% of Insilico Medicine’s resources. “He is utilizing AI to very rapidly accelerate drug discovery and development,” said Zhavoronkov. “That’s the reason why we managed to do nine preclinical candidates last year. We have one proven case where we discovered a new target that generated small molecules and went all the way to Phase I, and Phase II is ready to start in the United States.”

Zhavoronkov wants the best people to work at Insilico Medicine. “We’re super inclusive,” he said. “If you are an alien from a different planet and land in my backyard, come out of this flying saucer and give a hand, I will shake it and say welcome. We don’t care who you are as long as you are really good!”
He first started by hiring people through competitions on AI, such as taking the top three fastest people to outperform the latest release of Google’s DeepMind. With the displacement of people due to the war in Ukraine, at their Abu Dhabi location, Insilico has taken in about 65 “AI refugees” from this region.
Dust in the wind
Part of the legacy that Zhavoronkov wants with Insilico Medicine is that anyone can find drug targets, no matter where they’re from. They could be from countries that have never played a role in drug discovery, or they could be high schoolers. I know the latter case is possible because Zhavoronkov showed me a series of papers that were just published by three high schoolers in collaboration with Insilico Medicine using generative AI to help identify new therapeutic targets for glioblastoma multiforme and aging—his muse.
Like other ambitious biotech entrepreneurs and investors, Zhavoronkov is an aging research aficionado. His interest in aging was his jumping-off point for his entire career. While working a well-paying job in information technology in the early 2000s, he started to keep a pulse on aging research and realized that solving aging would require a computer scientist. So, he quit his job to enroll in the biotechnology program at Johns Hopkins University and then pursued a PhD in biophysics at Russia’s Moscow State University.
Next on Zhavoronkov’s list is to translate all of his work in AI and robotics into a clinical setting. “The patient sample would come in and would be processed in a very similar way, and you would get a prediction for the drugs that were FDA-approved that are good for this patient right there in the hospital,” he said. “If I do this in the next couple of years, I will be able to put a checkmark next to my life.”
While our interview was booked for 30 minutes, Zhavoronkov suddenly paused after an hour of talking. I don’t know why he stopped, although I did spy a notification that said to take a few minutes to himself. I probably couldn’t have stopped him earlier if I had tried. Zhavoronkov is a man on a mission who does not switch off. And I don’t think anyone can stop him.
NEW YORK and HONG KONG, March 13, 2024 /PRNewswire/ — There are thousands of diseases worldwide with no cure or available treatments. Traditional drug discovery and development takes decades and billions of dollars and more than 90% of these drugs fail in clinical trials. The emergence of artificial intelligence (AI) holds promise for streamlining and improving the entire process. However, ushering in a new era of AI-driven drug discovery requires costly and lengthy validation in preclinical cell, tissue, and animal models and human clinical trials.
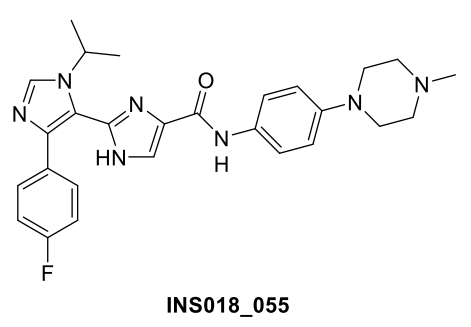
Now, that preclinical and part of that clinical validation was published in a new study in Nature Biotechnology. In this paper, Insilico Medicine and collaborators present the journey of its lead therapeutic program with an AI-discovered target and novel molecule generated from AI algorithms to Phase II clinical trials. For the first time, the paper discloses the raw experimental data and the preclinical and clinical evaluation of the potentially first-in-class TNIK inhibitor discovered and designed through generative AI. The study underscores the benefits of AI-led drug discovery methodology to provide efficiency and speed to drug discovery and highlights the promising potential of generative AI technologies for transforming the industry.
“When our first paper in the generative AI for generation of novel molecules was published in 2016, followed by many follow-up papers, the drug discovery community was very skeptical. Even after several validation experiments and launch of our AI software platform that is now used by many biopharma companies, many questions remained. Based on the research data, especially those from the live clinical program. To date, I have not seen anything close from any other company in our field,” said Alex Zhavoronkov, PhD, founder and CEO of Insilico Medicine. “From my perspective, the progress of INS018_055 has significant implications for the drug discovery field. It not only serves as a proof-of-concept for Pharma.AI, our end-to-end AI-driven drug discovery platform, but sets a precedent for the potential of generative AI to accelerate drug discovery. Using the publication as a guide, one can extrapolate how generative AI drug discovery tools may streamline early discovery efforts. We anticipate that the expanded application of this platform will address challenges facing industry R&D, including cost and efficiency, and accelerate the delivery of innovative therapies to patients.”
Insilico initiated the research by focusing on fibrosis, a biological process closely associated with aging. The group first trained PandaOmics, the target identification engine of Insilico’s proprietary AI platform Pharma.AI, on the collection of omics and clinical datasets related to tissue fibrosis. Next, PandaOmics proposed a potential target list using deep feature synthesis, causality inference, and de novo pathway reconstruction. After that, the natural language processing (NLP) models of PandaOmics analyzed millions of text files, including patents, publications, grants, and clinical trial databases to further assess the novelty and disease association. TNIK was identified as the most promising anti-fibrosis target. Notably, TNIK had been indirectly linked to multiple fibrosis-driven pathways in previous research but was never pursued as a potential target for IPF. In a separate paper, Insilico scientists demonstrated that TNIK may be implicated in multiple hallmarks of aging.
After selecting TNIK as a primary target, Insilico scientists utilize Chemistry42, the Company’s generative chemistry engine, to generate novel molecular structures with the desired properties using the structure-based drug design (SBDD) workflow. Chemistry42 combines over 40 generative chemistry algorithms and over 500 pre-trained reward models for de novo compound generation, and can optimize both generation and virtual screening based on expert human feedback. Following multiple iterative screens, one promising hit candidate demonstrated activity with nanomolar IC50 values. The group further optimized the compound to increase solubility, promote a good ADME safety profile, and mitigate unwanted toxicity while retaining its remarkable affinity for TNIK, which ultimately produced the lead molecule INS018_055, with less than 80 molecules synthesized and tested.
In subsequent preclinical studies, INS018_055 demonstrated significant efficacy in vitro and in vivo studies for IPF and showed promising results in pharmacokinetic and safety studies across multiple cell lines and multiple species. Furthermore, INS018_055 showed pan-fibrotic inhibitory function, attenuating skin and kidney fibrosis in two additional animal models. Based on these studies, INS018_055 achieved preclinical candidate nomination in February 2021, in less than 18 months following PandaOmics’ proposal of TNIK as a potentially novel target for IPF in 2019.
INS018_055 has exhibited excellent performance in clinical trials to date. In November 2021, 9 months after PCC nomination, the first healthy volunteers were dosed in a first-in-human (FIH) microdose trial of INS018_055 in Australia. This microdose trial exceeded expectations, delivering a favorable pharmacokinetic and safety profile that successfully demonstrated this clinical proof-of-concept and set the stage for the next step of clinical testing. In Phase I trials carried out in New Zealand and China, INS018_055 was tested in 78 and 48 healthy subjects, divided into cohorts focusing on a single ascending dose (SAD) study and multiple ascending dose (MAD) study. The international multi-site Phase I studies yielded consistent results, demonstrating favorable safety, tolerability, and pharmacokinetics (PK) profiles of INS018_055, and supporting the initiation of the Phase II studies.
“Combining AI methods with human intelligence, we have successfully nominated INS018_055, a potentially first-in-class antifibrotic inhibitor, with significant reductions in time and costs”, said Feng Ren, PhD, co-CEO and Chief Scientific Officer of Insilico Medicine. “Encouraged by positive preclinical and available clinical data, we look forward to favorable performance of INS018_055 in Phase 2 clinical trials, which would provide innovative options for patients while bringing more solid evidence for the AI-driven drug discovery industry.”
At the time of this publication, two Phase 2a clinical trials of INS018_055 for the treatment of IPF are being conducted in parallel in the United States and China. The studies are randomized, double-blind, placebo-controlled trials designed to evaluate the safety, tolerability and pharmacokinetics of the lead drug. In addition, the trials will assess the preliminary efficacy of INS018_055 on lung function in IPF patients. As this drug continues to advance, it drives hope for the roughly five million people worldwide suffering from this deadly disease.
Insilico’s drug discovery efforts are driven by its validated and commercially viable AI drug discovery platform, Pharma.AI, which works across biology, chemistry, and clinical medicine, providing the biotechnology and the pharmaceutical industry with advanced generative AI tools to accelerate their internal research and development. Powered by Pharma.AI, Insilico is delivering breakthroughs for healthcare in multiple disease areas, including fibrosis, cancer, immunology and aging-related disease. Since 2021, Insilico has nominated 18 preclinical candidates in its comprehensive portfolio of over 30 assets and has advanced six pipelines to the clinical stage.
Industry Commentary and Additional Information
“There has been much speculation that AI and deep learning methods will have a substantial role in shaping the future course of drug development. This paper presents a very convincing proof of concept.” says Charles Cantor, PhD, Scientific Advisory Board (SAB) at Insilico Medicine, “Driven by AI at nearly every stage from target identification to drug candidate selection, to phase 1 studies, a novel molecule is now ready for phase 2 clinical trials. If this process proves to be general, drug development without AI may well become inconceivable.”
“Healthcare is undergoing an important transformation of digitalization.” says Dr.Kai-Fu Lee Chairman of Sinovation Ventures, CEO of 01.AI. “I believe the use of AI and data science will revolutionize the field of medicine. Insilico Medicine’s TNIK program is a great example, presenting a breakthrough paradigm for discovering medicines from scratch under generative AI in chemistry and biology. The milestones achieved by Insilico, backed by compelling experimental data, will encourage the entire ecosystem that we are marching down the right track to advance life science with state-of-the-art information technology.”
“Although lots of companies are working on AI to improve different steps in drug discovery, Insilico is trying to apply their AI in all early drug discovery and design stages, which is so exciting to me,” says Michael Levitt, PhD, Nobel Laureate in Chemistry, 2013. “Insilico is literally AI from A to Z. They not only identified a novel target, but also accelerated the whole early drug discovery process and they’ve quite successfully validated their AI methods in the TNIK program. Drug discovery is a very wide-ranging project with a lot of uncertainty. AI can cope well with specific techniques for huge amounts of data, and by combining them with clever filtering, we can gain certainty and options from uncertainty.”
“Nowadays, it seems that we read about the virtues of AI, ML, generative design almost daily. There is a feeling that, perhaps, it is overhyped,” says Stevan Djuric, PhD, adjunct professor in medicinal chemistry at the University of Kansas and former head of the global medicinal chemistry leadership team at AbbVie. “However, in this paper, the Insilico Medicine team convincingly demonstrates the power of their proprietary platform which features target identification and validation, medicinal chemistry design and clinical trial components using the aforementioned tools. For experienced medicinal chemists, improving potency of compounds is often not the major challenge, but rather fine tuning of PK (CLu etc) and safety (off-target effects). The Insilico engine, in the case presented, successfully tackled all components of these difficult problems in a particularly timely manner. We will eagerly await news on the further progress of this agent and further clinical candidates discovered using generative AI paradigms.”
“Many people say they are doing AI for drug discovery,” says Alán Aspuru-Guzik, PhD, professor of Chemistry and Computer Science at the University of Toronto and director of the Acceleration Consortium. “A handful are delivering. Insilico’s team has shown both, the identification of a target followed by the development of a therapeutic agent all driven by AI. This, to my knowledge, is the first AI-generated drug in stage II clinical trials. A true milestone for the community and for Insilico. The lessons learned here can be further expanded to accelerate the discovery and development process.”
“When we started the preclinical profiling of INS018_055 for fibrotic diseases, everybody was seeing its target TNIK as a candidate for oncology indications,” says Klaus Witte, MD, German Medical Board certified Pharmacologist & Toxicologist, Preclinical Consultant to Insilico Medicine. “Although I was skeptical in the beginning, all data that we generated clearly supported Insilico’s prediction of anti-fibrotic efficacy. Looking at the broad set of preclinical and clinical data that is meanwhile available, I’m confident that INS018_055 could become a very valuable treatment option in pulmonary fibrosis and other fibrosis indications. Having helped to bring the compound to its present stage is something I’m really proud of.”
“As a clinician, I see firsthand the need for novel, effective treatments. This landmark work highlights the central role that AI can play in accelerating the path from discovery to treatment, reshaping our strategy against diseases that currently have limited therapeutic options,” says Prof. Christoph Kuppe, PhD, physician scientist at the RWTH Aachen.
“I was struck by the incredible progress Alex and Insilico have made in a single decade,” says Bud Mishra, PhD, professor of computer science at New York University. “The paper focuses on idiopathic pulmonary fibrosis, a disease with complex genetics involving multiple genetic mutations. They broke this complex problem down into two parts: selecting a target (namely, TNIK), and then guiding the drug discovery process by designing small molecules that would bind to that target and make it ineffective. The first part uses heuristics that are based on the scientific experiences accumulated in the past (the target must be novel, easy to understand in terms of interactions with known pathways, and follow the approaches used by others in guiding drug discovery and clinical trials in the past) and hence ideal for NLP using LLMs. The second part uses randomized heuristics to search and optimize over complex combinatorial spaces using DNNs, capable of dealing with naturally occurring ‘easy instances of hard problems.’ Speculatively, the first part will become more difficult over time (hallucination vs. true novelty) and the second part, simpler, as Moore’s law will continue to improve the computational power exponentially.”
Reference
Ren, F., et al. A small-molecule TNIK inhibitor targets fibrosis in preclinical and clinical models. Nat Biotechnol (2024). https://doi.org/10.1038/s41587-024-02143-0