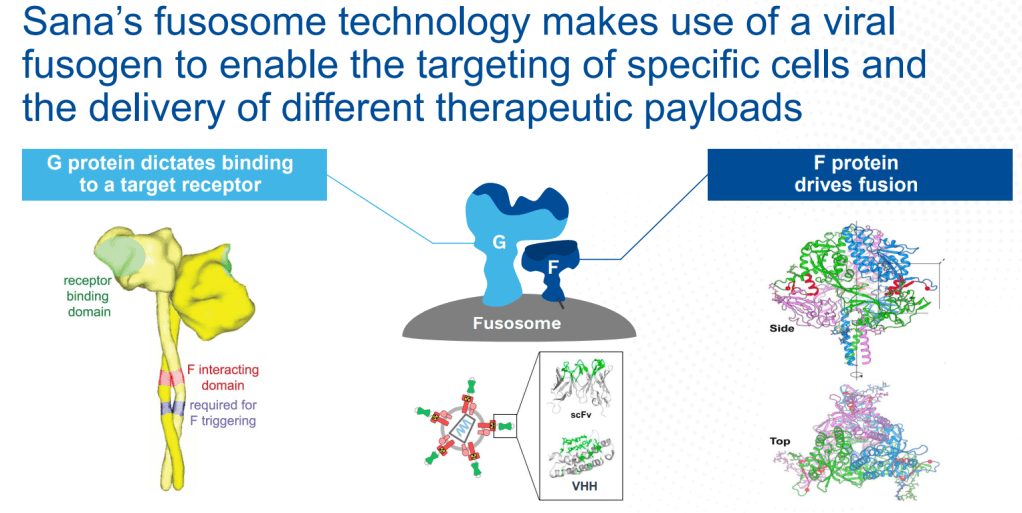
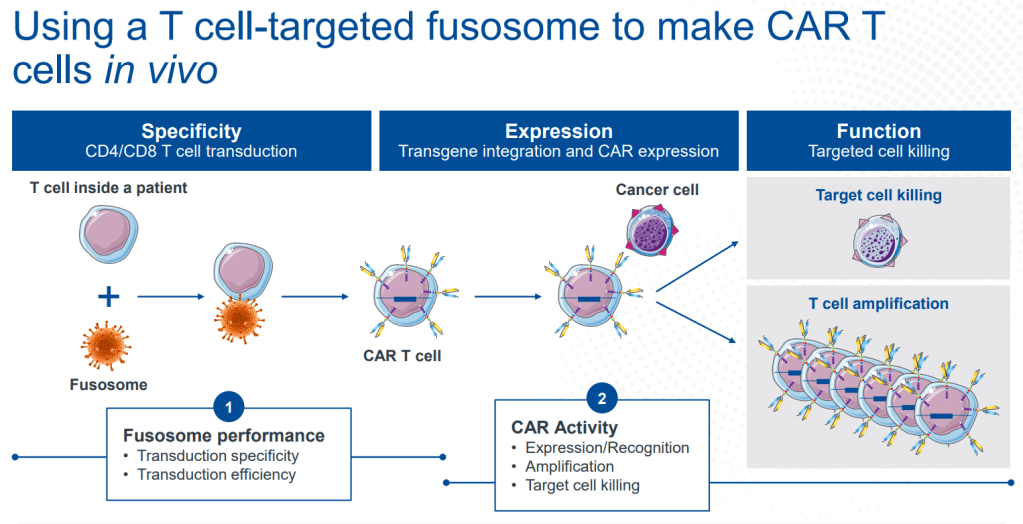
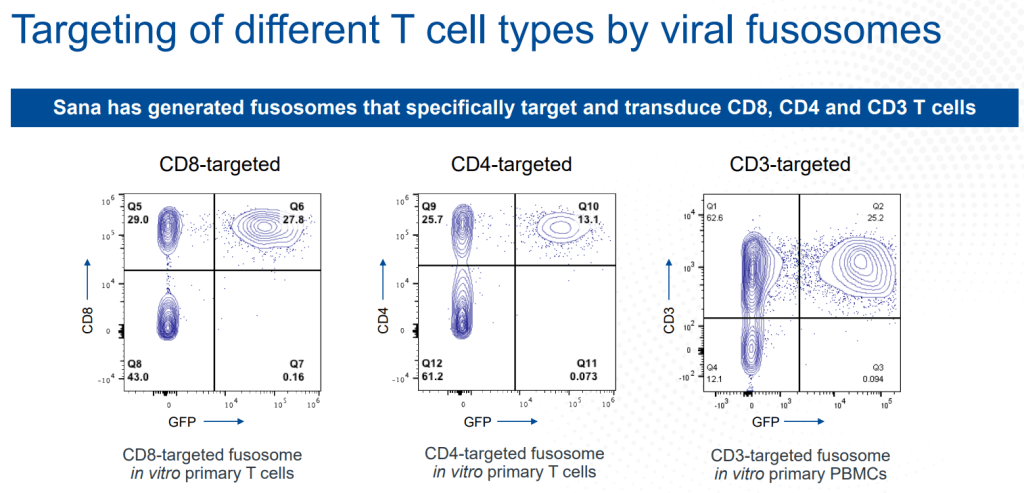
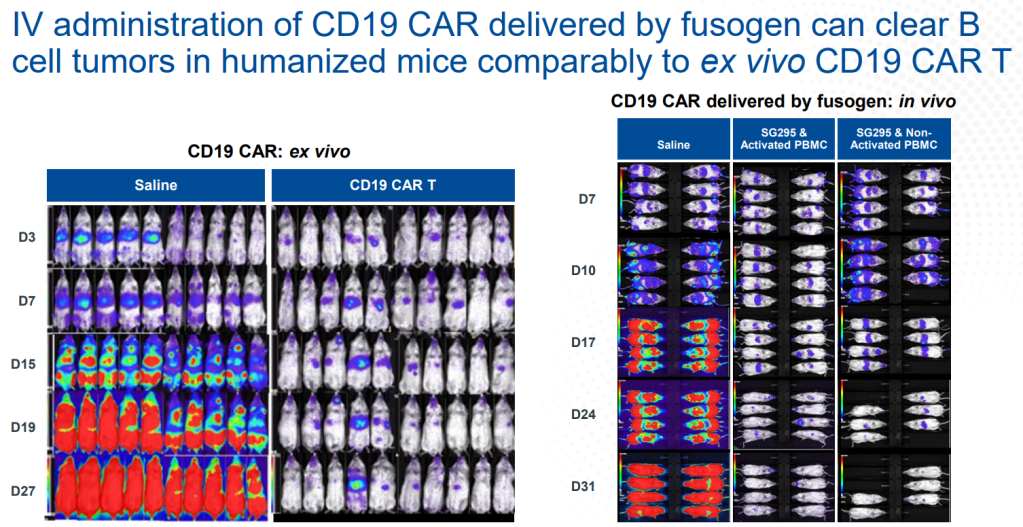
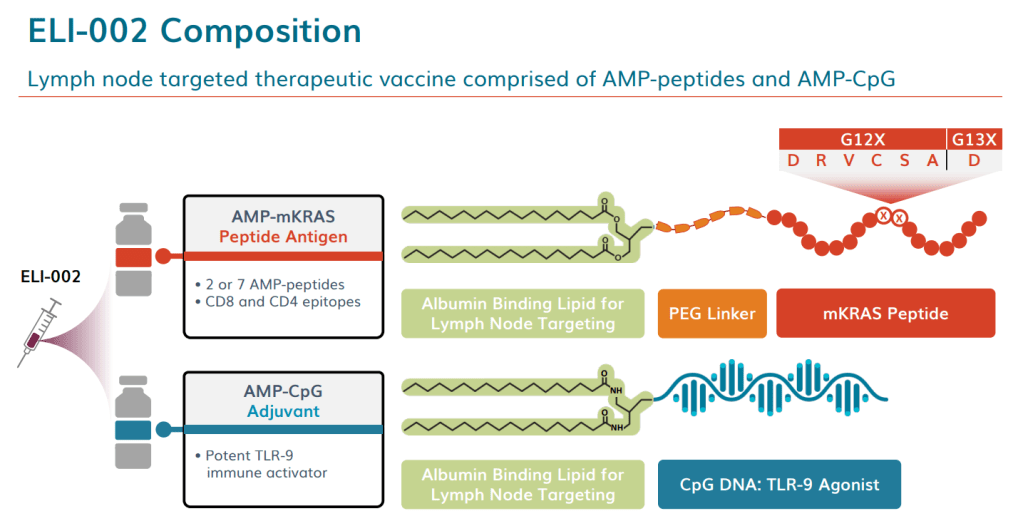
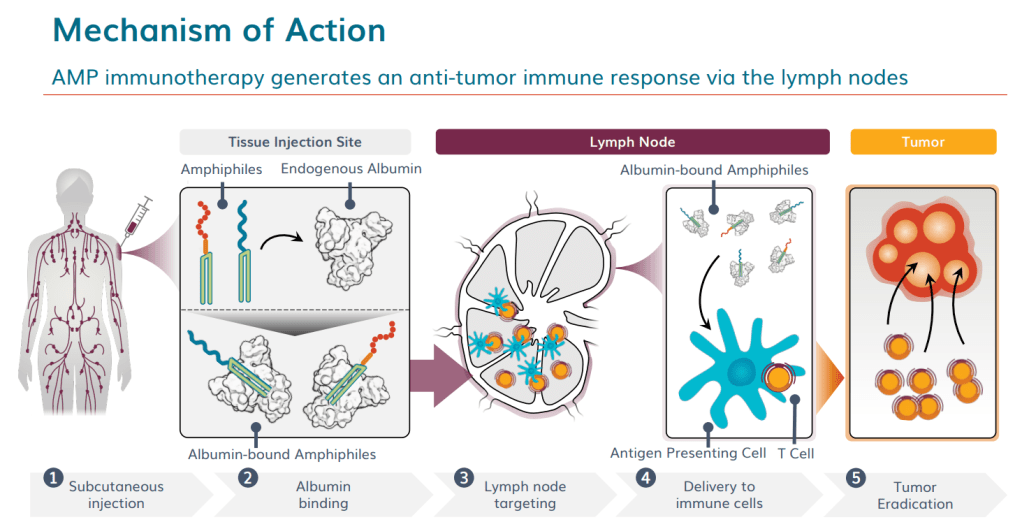
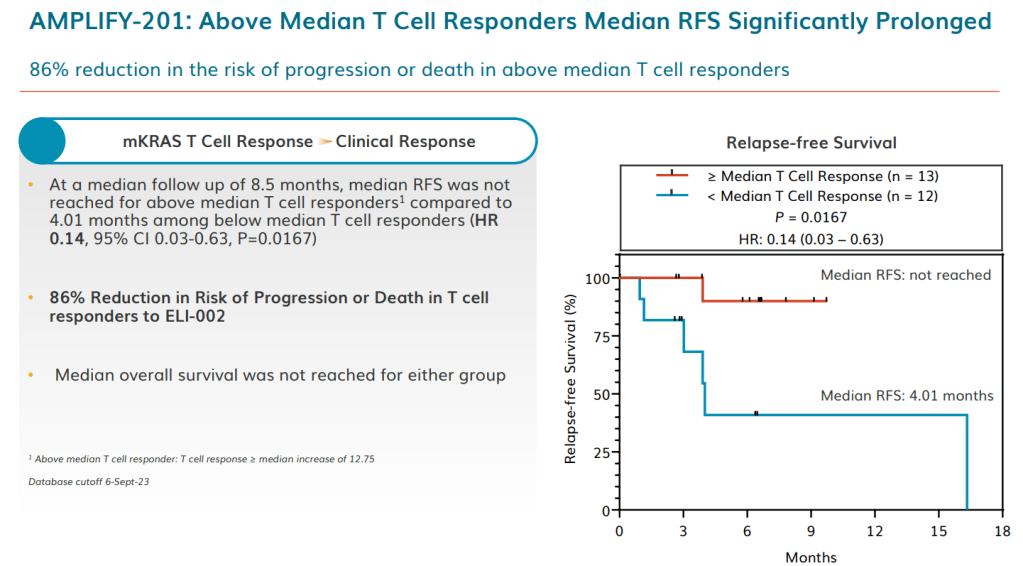
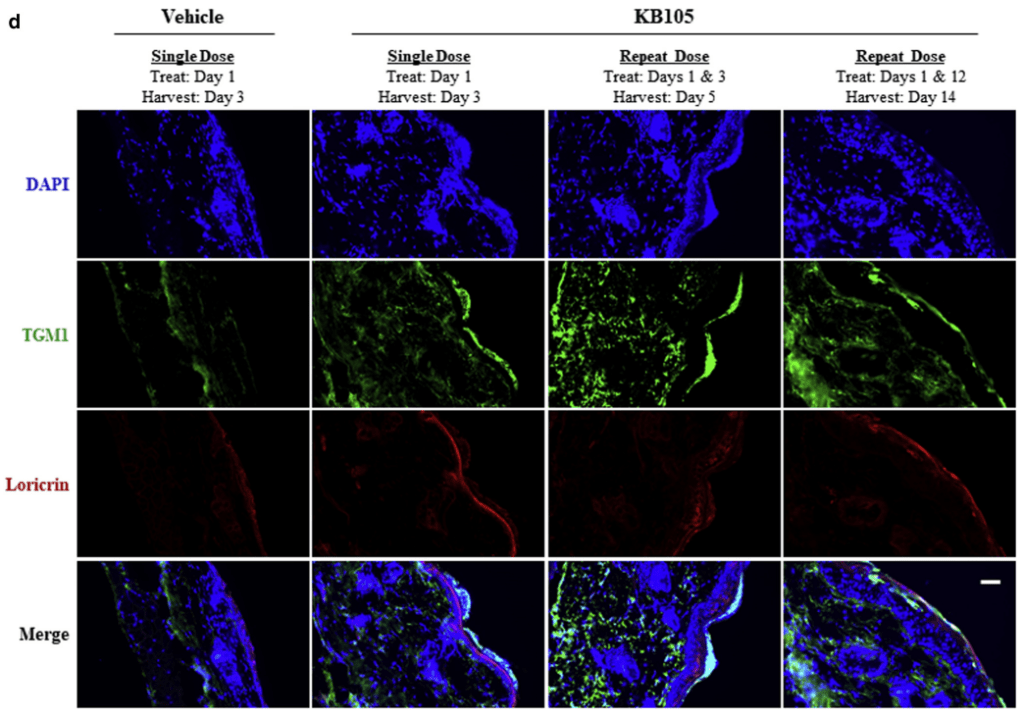
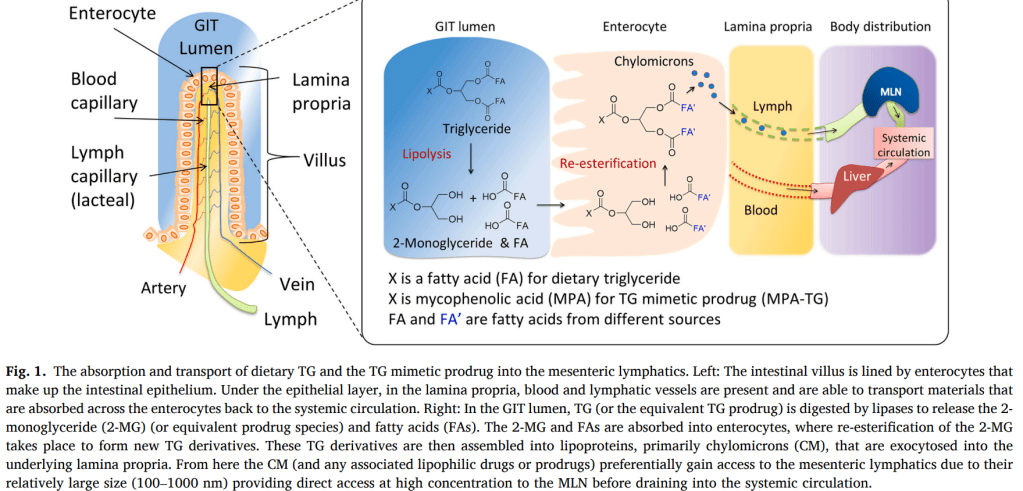
안녕하세요 보스턴 임박사입니다.
Greg Verdine박사는 Harvard University교수이면서 VC이고 Serial Entrepreneur인 특이한 경력을 가지고 있는데요. 예를 들면 Wave Life Sciences에서 WaVe의 Ve는 Verdine의 Ve에서 따온 것입니다. 앞의 Wa는 Wada 교수의 Wa에서 따온 것이고요.
BIOTECH (8) – Wave Life Sciences의 RNA Editing 신약 가능성에 GSK가 투자하다.
Greg Verdine교수의 연구 중 가장 중요한 연구는Stapled Peptides라는 분야인데 이것은 Protein의 alpha Helix를 Chemical Bonding으로 Stapling하는 것입니다. Aileron Therapeutics라는 회사를 만들어서 RCM (Ring Closing Metathesis) 방법으로 만든 Stapled Peptides Platform을 임상에 진행시켰으나 그리 성공적이지 못했습니다.
Despite treatment with the chemoprotective agent ALRN-6924, patients with p53-mutated breast cancer receiving neoadjuvant or adjuvant therapy with docetaxel, doxorubicin, and cyclophosphamide experienced grade 4 neutropenia and alopecia in a phase 1b trial (NCT05622058), failing to meet the trial’s primary and secondary end points of duration and incidence of severe neutropenia in cycle 1 and incidence of chemotherapy-induced alopecia, respectively.1 Based on these results, the company has decided to terminate the trial and further development of ALRN-6924, which was also under evaluation as a chemoprotective agent in p53-mutated small cell lung cancer and non–small cell lung cancer (NSCLC).
The company announced that it is exploring strategic alternatives in conjunction with Ladenburg Thalmann & Co., Inc., which may include an acquisition, a merger, a business combination, or a sale of assets or other transactions. Further statements from the company will not be released unless or until its Board of Directors has approved a definitive course of action or it is determined that other disclosure is appropriate.
현재는 Lung Disease로 Business Model을 완전히 바꾼 상태입니다.
Aileron Therapeutics (ALRN) Announces Acquisition of Lung Therapeutics – Street Insider 10/31/2023
Aileron Therapeutics의 Stapled Peptides 임상을 통해서 그래도 배운 것이 있을 것이라 생각합니다. Aileron을 설립한지 몇년 후에 다른 Stapled Peptides Platform 회사인 Fog Pharma를 2016년에 설립했는데요 Greg Verdine 교수연구실에서 박사학위를 받고 Postdoctoral Fellow로 있던 John McGee 박사가 하던 Cysteine Stapling을 상용화하기 위한 회사입니다.

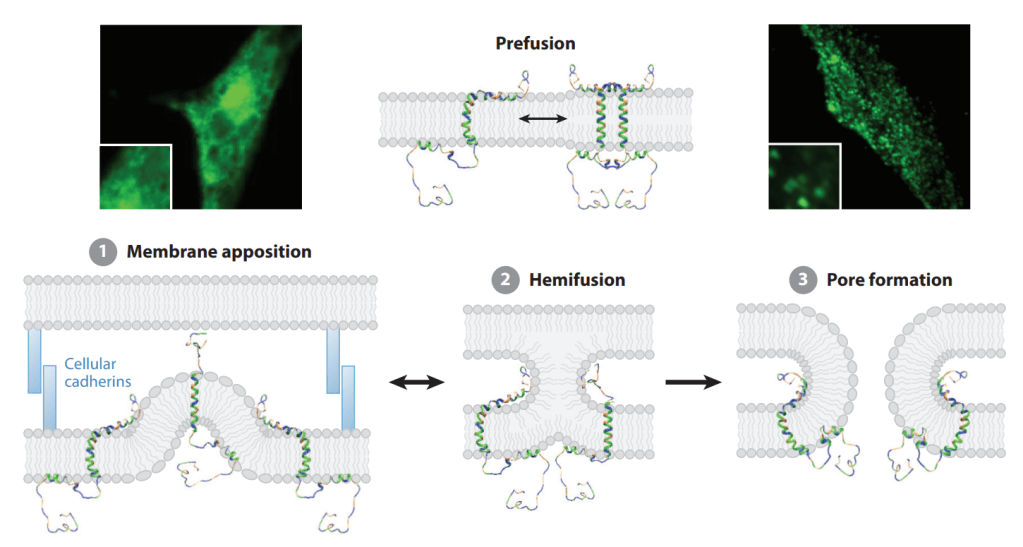
Cysteine Stapling은 아래와 같이 Internal Cysteines을 Alkylation으로 Stapling 하는 것입니다.

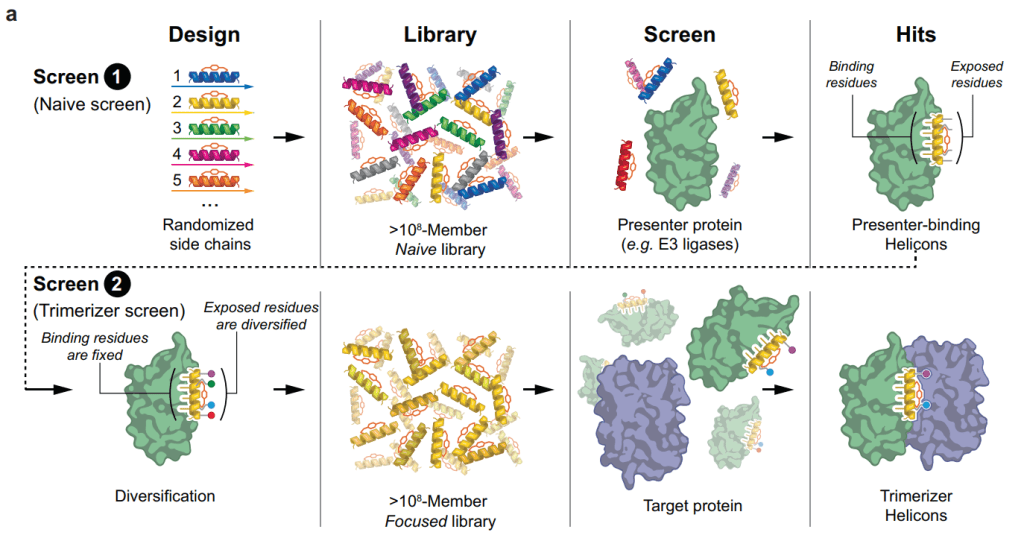
이 Stapled Peptides를 Ligand로 해서 Phage display로 High throughput screening을 합니다.

이 논문에서는 1차 agnostic naive screening을 한 다음에 hit을 가지고 2차 focused screening을 거쳐서 비교적 단기간에 Lead Candidates를 찾는 방법을 보고했습니다.
$66 Million Series B를 하고 Aileron과 달리 바로 임상에 진입할 Lead를 발표했습니다.
FogPharma Secures $66 Million Series B Financing. – PR Newswire 5/16/2019
FogPharma’s drug discovery engine has been configured to deliver multiple new medicines in rapid succession, with clinical entry for the first product, a first-in-class beta-catenin antagonist, by the end of 2019, followed by a steady stream of first-in-class clinical product candidates addressing other intractable targets.
그리고 2년 후에 $107 Million Series C를 했습니다. Pipeline도 늘어났습니다.
FogPharma’s proprietary hyperstabilized α-helical peptides (Helicon™ peptides) are a new class of therapeutics that combine the targeting strength and specificity of antibodies with the broad tissue distribution, intracellular target engagement and oral dosing optionality of small molecules. The Company’s Helicon peptide drug discovery engine integrates directed evolution, proprietary helix hyperstabilization chemistry, highly multiplexed drug optimization technology, artificial intelligence including deep learning and machine learning, structure-based drug discovery, and multiscale manufacturing to rapidly discover Helicon peptide therapeutics against important, previously intractable targets with broad applicability to virtually all disease areas.
- The Company’s first-and-only-in-class direct β-catenin inhibitor. Dysregulation of the Wnt/β-catenin signaling pathway has been shown to occur in at least 20% of all human cancers, with the true patient population likely being higher. FogPharma’s lead antagonist has been shown to surgically disrupt the interaction of β-catenin with its downstream transcription factor, TCF, and thereby disrupt signal transmission thorough the oncogenic arm of the Wnt pathway.
- A first-in-class YAP/TAZ-blocker TEAD antagonist, which is the only molecule presently in development that binds the fully activated form of TEAD. The YAP/TAZ-TEAD interface is part of the hippo pathway, where dysregulation has also been shown to occur in many cancers.
다음해에 다시 $178 Million Series D를 했구요. FOG-001의 임상을 2023년 중순에 시작하겠다는 계획을 발표했습니다.
FogPharma’s lead Helicon polypeptide development candidate, FOG-001, a first-and-only-in-class direct TCF-blocking β-catenin inhibitor with potential applicability to significant cancer patient populations, is expected to enter clinical development in mid-2023. In addition, FogPharma is advancing other first-in-class programs against important, biologically validated cancer targets that have remained elusive to other approaches including TEAD, NRAS, Pan-KRAS, ERG and Cyclin E1.
그리고 얼마전에 $145 Million Series E를 했구요. CEO를 J&J 출신인 Mathai Mammen으로 교체하면서 J&J ex-CEO Alexis Borisy가 투자자를 유치하고 BOD member로 들어오게 되었습니다.
Johnson & Johnson’s former pharma R&D chief, now the CEO of FogPharma, has brought on former J&J CEO Alex Gorsky as an investor in the company’s $145 million series E. Gorsky joins a syndicate of heavyweights like RA Capital and General Catalyst who became first-time Fog investors.
Returning investors included Arch, GV and Fidelity. Another big name on the bill is serial biotech entrepreneur Alexis Borisy, who also joined the board on behalf of Nextech, which led the round.
FOG-001 is an intracellular TCF-blocking β-catenin inhibitor being tested in a phase 1/2 trial for solid tumors. Mutations of the Wnt/β-catenin pathway, which the drug targets, are particularly common in colorectal cancer.
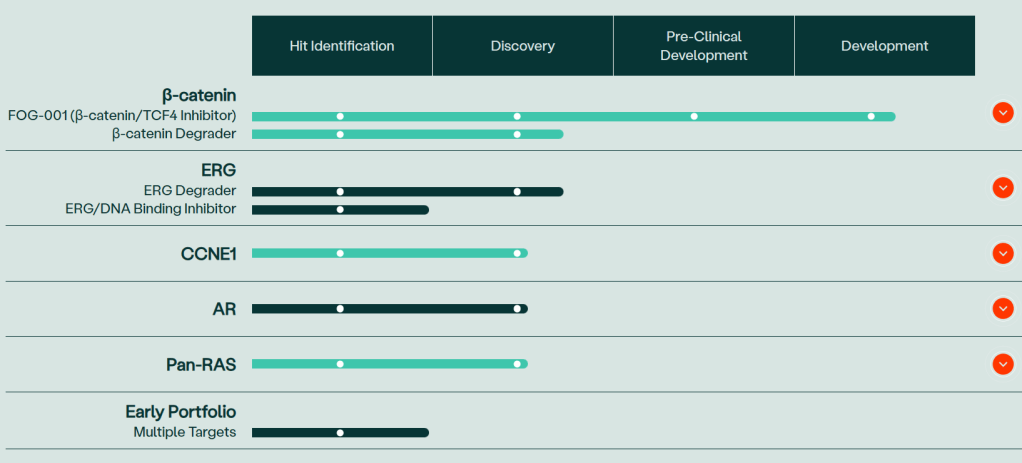
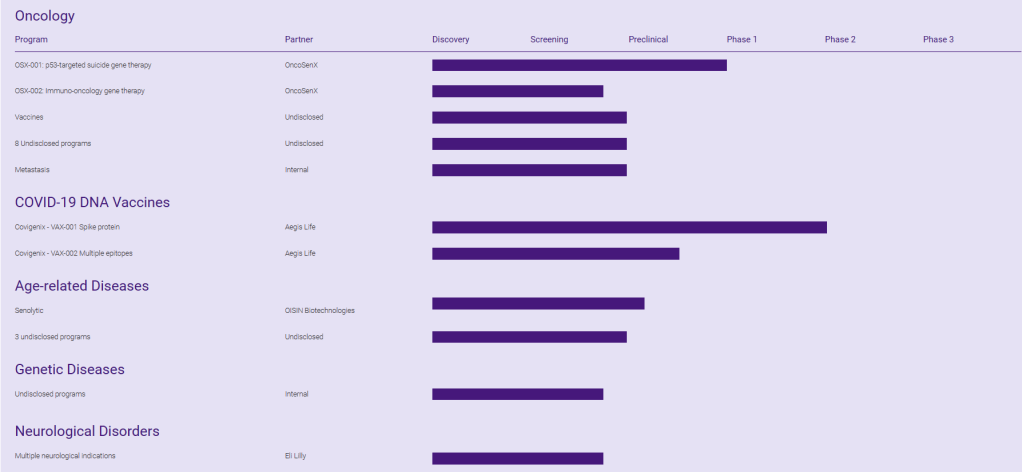
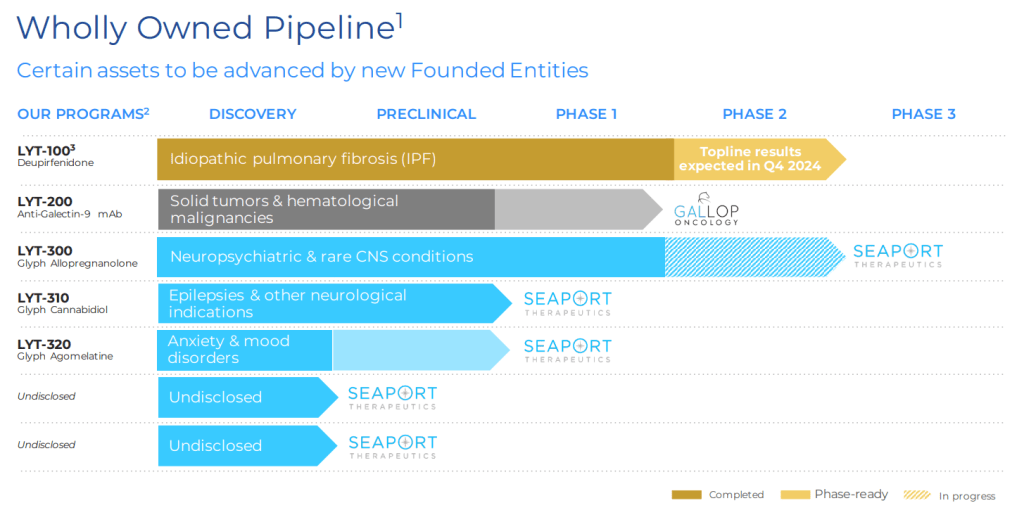
현재 Pipeline은 아래와 같습니다. FOG-001의 임상1/2상 데이타가 어떻게 나올지 궁금합니다.